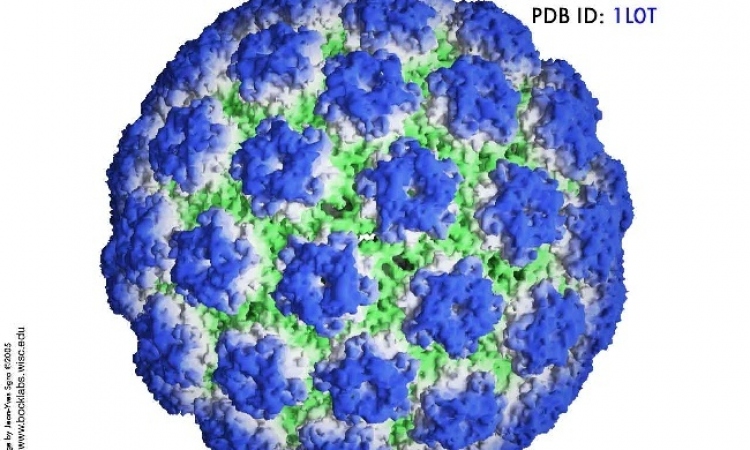
Papilloma virus infection
The latest analysis of a vaccine’s safety,
efficacy and immunogenicity indicates that
Cervarix, a human papilloma virus (HPV)
vaccine offers sustained protection for over
six years against infection by viral types
HPV-16 and HPV-18, most commonly associated
with cervical cancer.
Published in an Article Online First (www. thelancet.com) and in The Lancet, the study was led by Dr Cosette Wheeler (New Mexico University, Health Sciences Centre, Albuquerque, NM, US A) and colleagues from the GlaxoSmithKline Vaccine HPV-007 study group.
Findings from several clinical studies have shown that Cervarix, the HPV-16/18 AS04- adjuvanted vaccine, induces a strong and sustained antibody response and has a favourable safety profile. The present study consisted of an initial efficacy study, which began in 2001, and a long-term follow-up study in 2003. Results of the initial study and two interim analyses of the follow-up study have been reported previously. Here, the authors report the final analysis of the follow-up study, with a total follow-up of up to 6.4 years after vaccination.
The original study analysed 1,113 women aged 15–25 years, with a normal cervical profile, who had no evidence of infection with HPV-16 or 18 or any of 12 other cancer-causing HPV types. 27 sites in three countries participated in the followup study, which included 393 women in the vaccine group and 383 in the placebo group. Cervical samples were tested every six months for HPV DNA. The primary objective was to assess long-term vaccine efficacy in the prevention of incident cervical infection with HPV 16 or HPV 18, or both. The authors performed a combined analysis of efficacy data from the initial and follow-up study up to 6.4 years after first vaccination, in order to give a good estimation of the overall vaccine efficacy, both short-term and long-term.
Vaccine efficacy against incident infection with HPV-16/18 was 95% and against 12-month persistent infection was 100%. Vaccine efficacy against CIN2+ was 100% for lesions associated with HPV-16/18 and 72% for lesions independent of HPV type. Antibody concentrations (against HPV- 16/18) remained at least several fold higher in vaccinated individuals than would be found after natural HPV infection. Safety outcomes were similar between groups: during the follow-up study, 30 (8%) participants reported a serious adverse event in the vaccine group versus 37 (10%) in the placebo group. None was judged to be related, or possibly related to vaccination, and no deaths occurred.
The authors say: ‘The study population was continuously exposed to HPV infections, as indicated by the continuous accrual of CIN cases in the placebo group, showing that the vaccine confers sustainedprotection and that efficacy does not wane up to 6·4 years after first vaccination. Vaccine efficacy against persistent infection with HPV-16/18 remained 100%.’ The authors also point out that vaccine efficacy was achieved against incident infection with HPV-31 and HPV-45, two cancercausing HPV types related to HPV-16 and HPV-18, respectively. They said: ‘HPV-31 and HPV-45 are among the types most frequently associated with cervical cancer after HPV-16 and HPV-18, and are responsible for 10% of all cervical cancer cases.’
In terms of public health benefit, the authors say that adolescents before sexual debut are the main target for immunisation because they are most likely to benefit from population-based HPV vaccination programmes. They point out that one strength of their study is that it was done in women who were naive to cancer-causing HPV infection at the time of vaccination, and thus it attempts to represent the target population. Because the incidence of cervical cancer peaks on average more than 30 years after adolescence, the vaccine has to confer protection for many years. So far, this study shows the longest duration of protection against HPV-16 and HPV-18 infections for a licensed prophylactic HPV vaccine. Protection with the alternative HPV-6/11/16/18 vaccine has (so far) been shown for up to 5 years after vaccination.
The authors conclude: ‘Although further assessment is necessary to confirm longterm vaccine effects, in view of the data from our study, we expect protection to continue for many more years.’ In an accompanying Comment, Dr Gary M Clifford, of the International Agency for Research on Cancer, in Lyon, France, said that HPV vaccination is being considered by the GAVI Alliance for sustainable funding.
26.01.2010