© Toyohashi University of Technology. All rights reserved
News • Integration of microanalysis chips
Establishing microfluidic control technology for blood testing devices
Researchers in Japan have achieved major progress toward practical application of compact blood analyzers.
A research group supervised by Assistant Professor Shunya Okamoto of the Department of Mechanical Engineering, Toyohashi University of Technology, and another supervised by Associate Professor Yoshiaki Ukita of the Graduate Faculty of Interdisciplinary Research, Faculty of Engineering, University of Yamanashi, established a microfluidic control technology that can be applied to small blood testing devices and consequently developed a novel integrated immunoassay device. This microfluidic control technology is expected to address cost issues—one of the limitations to the social implementation of microfluidic chips—and realize a convenient blood testing device. The results of this study were published online in the journal RSC Advances.
Recommended article

Article • More than just a subdiscipline
Spotlight on clinical chemistry
Clinical chemistry has evolved far beyond its beginnings of blood and urine analysis. Advanced techniques such as enzyme measurement, electrophoresis, and immunoassays have made their impact on the field, expanding its diagnostic capabilities. Keep reading for advances in automation and clinical chemistry applications for toxicology, urinalysis and much more.
Compared with conventional analysis systems, analysis systems using microfluidic chips offer advantages such as minimized sample volume and shortened analysis time. However, a technology that precisely controls fluids in extremely minute spaces of approximately 50–300 μm is required. Enzyme-linked immunosorbent assay (ELISA) is an analysis technique used in blood analysis for measuring cancer biomarkers; however, although highly sensitive, ELISA requires complex fluid control.
Conventional studies have employed complex structures and operations, such as placing numerous valve structures and connecting pumps, to perform such fluid control, thus increasing the cost of devices and test chips; moreover, extensive user training is required to operate such devices. These issues have become major limitations to the social implementation of ELISA.
To address these issues, this study developed a centrifugal microfluidic chip with autonomous control functions. The chip comprises a microfluidic chip with only a simple channel structure that can be molded using mass-producible technologies such as injection molding. Further, the researchers perform analysis by simply rotating the chip at a steady rotation speed for several tens of minutes (steady rotation). Thus far, the team has demonstrated that this autonomous-controlled centrifugal microfluidic chip can perform ELISA, which requires complex fluid control. However, the main limitations are that numerous samples are required for analysis, and users require multiple chips and extensive dispensing operations to quantify the test substance.
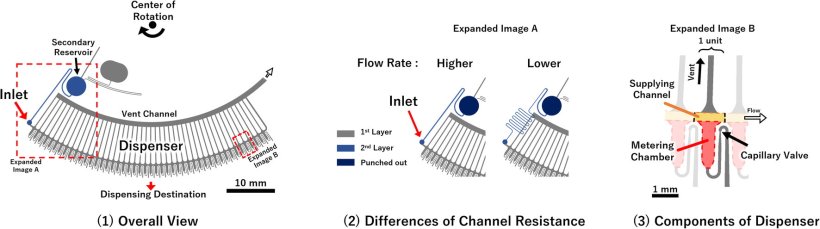
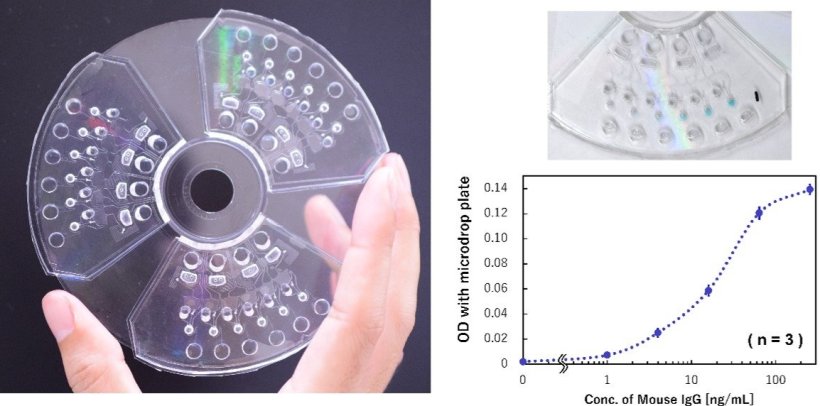
Image source: Okamoto S, Nagai M, Shibata T, Ukita Y, RSC Advances 2024 (CC BY 3.0)
To address these limitations, the researchers developed a dispensing mechanism that can automatically distribute reagents instead of manual labor and demonstrated the operation of a "multi-sample microvolume simultaneous analysis device" that implements the dispensing mechanism in an analysis unit. The analysis demonstration conducted using IgG antibodies as test substances showed that although the sample volume was sufficiently small—approximately 5 μL (mm³), which can be collected even by finger prick—and has the same detection sensitivity (63.4 pg/mL) as that of the previous manual analysis, the analysis time can be shortened by one-third to approximately 30 min.
"Our research results use prototype devices constructable at the laboratory level. However, in the future, we will investigate practical applications by optimizing the design using mass-producible processing methods and materials", the researchers state, adding that this fluid control technology makes it possible to realize control with microfluidic chips with a simple structure. In addition, the centrifugal control device is also simple to operate because fluid control is feasible at a steady rotation speed. Therefore, this technology can contribute toward addressing cost issues—one of the limitations of the social implementation of microfluidic chips. Currently, blood analyzers are installed only in central laboratories of general hospitals. However, with advances in cost reduction and device miniaturization, along with improved operability, blood analyzers can be installed in clinics, drugstores, and households, thus rendering blood tests accessible and improving people's quality of life, the researchers conclude.
Source: Toyohashi University of Technology
29.05.2024