High-tech plaster for analgesic therapy
It sticks to the skin like a plaster, but this credit-card sized innovation contains an entire patient-controlled drug-delivery system.
Developed for post-operative pain management, Ionsys, manufactured by Jannsen-Cilag, was the product highlight at the 53rd German Anaesthesia Congress, in Leipzig.
Intravenous patient-controlled analgesia systems (IV PCA) are the current gold standard for post-operative pain management. This pre-programmed and electronically controlled pump system, called a ‘pain pump’, allows the patient to push a button to intravenously administer small doses of analgesic. The patient can repeat this procedure as often as he wishes, until an acceptable level of medication has been reached. Certain risks are associated with this system, in particular incorrect programming of the machine, an incorrect analgesic solution, or problems with the intravenous access.
A new procedure aims at reducing these risks: the patient-controlled transdermal analgesia (PCTA) system. The credit-card sized system is applied to the patient’s arm or chest and functions like an IV PCA – however without an IV access. The system comes pre-filled with the analgesic fentanyl and is pre-programmed. By pushing a button, 40 micrograms of the medication are released, using a low-intensity electrical current of 170 mA (so-called iontophoresis), and the dose is transported through the skin over a period of ten minutes. During that time, the system does not respond to further prompts for medication release. Thus an increase of the dosage by the patient is excluded. 80 doses total can be administered. The number of doses administered is identified semi-quantitatively and displayed via an LED.
PCTA has been proven to be effective and safe compared with a placebo (Chelly JE, Grass J, Houseman TW, Minkowitz H, Pue A (2004) The safety and efficacy of a fentanyl patient-controlled transdermal system for acute postoperative analgesia: a multicenter, placebo-controlled trial. Anesth Analg 98:427-433) and compared with the standard procedure the patient-controlled intravenous analgesia (Viscusi ER, Reynolds L, Chung F, Atkinson LE, Khanna S (2004) Patient-controlled transdermal fentanyl hydrochloride vs intravenous morphine pump for postoperative pain. JAMA 291:1333-1341).
Stefan Grond, Professor of Anaesthesiology at Martin Luther University in Halle, Germany, considers this system ‘…an important step towards empowerment of the patient’. Nurses and physiotherapists will also welcome this innovation because it facilitates and increases mobility of the patients - since an infusion pump is no longer required.
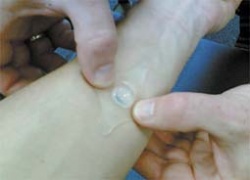
Vita-Clip - developed and produced by Serumwerk Bernburg - is another innovative plaster that can simplify the work of personnel. This plaster induces external vascular occlusion and consists of a silicone support, with a silicone lens in the centre and a reservoir under this, with a foil at the base, to which skin adhesive has been applied.
The skin is no longer directly punctured - but the silicone lens. If the needle is removed at the end of treatment, the reservoir under the lens fills up with a little blood from the puncture canal. This causes an extracorporeal haematoma, which presses the foil against the skin and stops bleeding, without having to be pressed by hand. Professor Reinhard Weidhase, Manager for Innovation and Registration, considers this to be particularly advantageous for dialysis patients.
The silicone lens increases the visibility of the puncture site, so that a dialysis shunt is hit more precisely and not damaged. The puncture needle is stabilised, reducing pain. Subsequent bleeding from the shunt is reliably avoided. The side of puncture remains sterile for a longer period and infections are reduced. Last but not least, time is saved, as neither the personnel nor the patient has to apply pressure by hand. The first users have reported that the skin tolerability is very good, particularly when it is thin and atrophic. They say that it is very comfortable to wear, as they can take a shower or bath after dialysis (Source: Dialyse aktuell 2006; 10 (3): 52-53).
30.08.2006