Gastric bypass undoes diabetes symptoms
French findings support the notion that gastric bypass can rapidly reverse diabetes symptoms' a side-effect that lap-band surgery does not show. The benefit results from a new-found element of glucose production by the intestine which also increases insulin sensitivity and lowers blood sugar.
The insights could be helpful in treating obese patients in particular but might even have potential for people who are diabetic, whether they are obese or not.
The report in the September Cell Metabolism, a publication of Cell Press, shows by studying mice that have undergone both procedures - gastric bypass and lap-band surgerg - that changes in the intestine are the key.
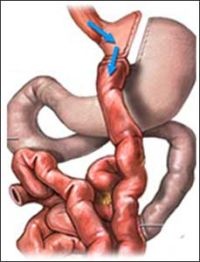
In addition to removing about two-thirds of the stomach, gastric bypass in effect produces a "double intestine," said Gilles Mithieux of Institut National de la Sante et de la Recherche Medicale in France. The portion closest to the stomach is taken out of the loop so that it receives no nutrients. The segment normally farther down is then attached directly to the stomach, where it receives all the nutrients coming in.
In both cases, those positional changes ramp up production of blood sugar by the small intestine, Mithieux said. He noted that fasting normally induces blood sugar production by the upper small intestine. By placing the lower small intestine, which doesn't normally produce much glucose, in close proximity to the stomach, it starts to act more like the upper portion.
That blood sugar synthesized in the intestine pours into the portal vein (a large vein that carries blood from the digestive tract to the liver) where it sends a signal to the brain, he and his colleagues earlier found. "The walls of the portal vein system detect the glucose and inform the brain," he said. "It's an important signal for decreasing hunger."
They now find an important new element of glucose production by the intestine. It also increases insulin sensitivity and lowers blood sugar, improving the symptoms of diabetes. Mithieux said that's in part because glucose production by the intestine lowers glucose production by the liver, which accounts for a much greater overall proportion of blood sugar synthesis. These metabolic changes take place within days of surgery, well before any weight loss takes place.
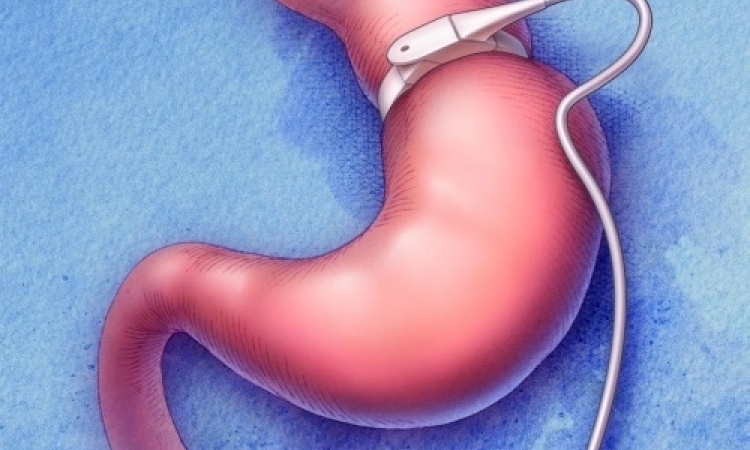
While gastric banding, in which a prosthetic band is placed around the upper stomach, works for weight loss, it doesn't affect the intestine at all and lacks the immediate metabolic benefits of bypass, they report.
Further evidence for the mechanism involved came from studies of mice lacking GLUT-2, a glucose transporter required for glucose sensing in the portal vein. Gastric bypass lost its insulin-sensitizing benefits in those GLUT-2-deficient mice. Similarly, mice whose portal veins had lost their nerve supply also stopped responding to the surgical procedure.
The bottomline, according to Mithieux: the intestine deserves more respect. "Up to now, the intestine had been considered like a machine to assimilate nutrients. We've now begun to realize that it is a complex endocrine organ" with particular importance when it comes to glucose metabolism.
As for what patients weighing gastric bypass versus lap-band surgery should do, Mithieux recommends they talk to their doctors about the possible benefits and risks of both procedures.
Journal reference: Stephanie Troy et al. Intestinal Gluconeogenesis Is a Key Factor for Early Metabolic Changes after Gastric Bypass but Not after Gastric Lap-Band in Mice. Cell Metabolism, September, 2008
04.09.2008