Source: Shutterstock/kentoh
Article • Pushing for integration with radiology
Digital pathology & AI: Multiple viewing improves consultation
Pathology is embracing digitisation just as radiology did over 20 years ago and both specialties are looking at new ways to integrate each other in their workflows. AI could fuel this sparkling alliance, giving it further means to improve cancer treatment.
Report: Mélisande Rouger

A panel of experts from both disciplines spoke about the developments and opportunities at ECR 2019. Radiologists and pathologists both work with images and have very similar workflows, according to Lluís Donoso Bach, Director of the Diagnostic Imaging Department at Hospital Clínic of Barcelona. ‘We both perform image acquisition, visualisation and then write a report. Digital images can now also be viewed by pathologists. Exploring new cooperation paths is a natural evolution,’ Bach explained. Benefits that can emerge from increased cooperation between the two disciplines include immediate access to data needed in clinical routine, which can become handy in multidisciplinary tumour boards and for remote physicians, and avoid the risk of breaking, losing or wrongly sorting glass slides.
In prostate biopsy, we have to estimate the volume that’s been taken up by the tumour. How do you do this is in a non-digital way? It’s just completely not reproducible and inaccurate
Paul J van Diest
For the past eight years, Paul J van Diest, head of the Pathology Department at the University Medical Centre (UMCU) in Utrecht, has successfully worked to digitally transform his department. His team now has a complete digital pathology workflow, which considerably improves efficacy, he said. ‘There is no more case assembly in the lab, but quicker diagnostics and preparation of multidisciplinary meetings. Searching slides for a big oncology meeting would take a full day. Today, there’s no more of that; everything is on our screen. We hardly ever go to the archive anymore.’ Working in a digital environment also enables annotations to be taken, notably 3-D annotations, which is much easier and safer than writing on slides. ‘We used to scribble with a pen on slides. Now we do annotations like radiologists have been doing for decades. It’s so much easier, for example when taking measurements to the resection margins. In prostate biopsy, we have to estimate the volume that’s been taken up by the tumour. How do you do this is in a non-digital way?’ he asked. ‘It’s just completely not reproducible and inaccurate.’
Probably the best selling point of digital pathology is that it improves patient safety. The list of benefits is long: easier and more accurate measurements and counting; better overview of tissue present; the possibility to flag missing slides, track viewing and automatically link images to reporting and the LMS. ‘This means there’s no more wrong report to the wrong patient – no more mixing up of slides,’ van Diest said.

Switching to digital also means multiple observer viewing, which improves workflow and consultation. Revision between labs can also be done on the same day and pathologists can work paperless and with better ergonomics. Teaching is also much easier, since students like digital teaching more than slides. Van Diest is now working on integrating images into the electronic patient record, another step that would improve workflow.
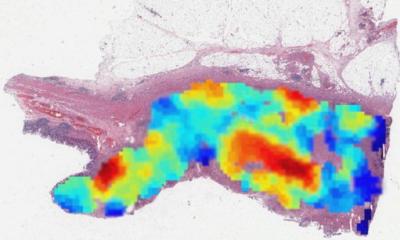
Integration with radiology makes more and more sense for pathologists and radiologists to improve the quality of their services and workflows. Digital pathology and AI are strongly pushing in that direction, van Diest argued. ‘There is definitely a case for integration of image analysis and deep learning. During diagnostic work, we often use completely subjective features to make a diagnosis. For example, we diagnose cancer when we see, within the tissue, big nuclei, variation of shape or size between the nuclei, or when the chromatic pattern of nuclei are irregular, etc. But these are all subjective, and we’re easily tricked in our visual system in assessing size, so we’re going to measure it,’ he explained.
Ideally pathologists could use AI and image analysis in several applications, e.g. mitoses, nuclear area, degree of tubule formation and metastases finder, ‘a tedious task, where we might miss things.’ Integration would also make sense in immune infiltrate quantification; IHC membrane scoring and positive nuclei scoring; and tumour vs. stroma quantification, which is becoming quite popular in breast and colon cancer. Mitoses counting in breast cancer usually takes pathologists 10-15 minutes. An AI algorithm could be used as a pre-screener that finds candidate mitoses to expedite the process. ‘We could click on the ones we like and don’t like and end up with a number of mitoses for a different area, of typically two square millimetre.’ In nuclear segmentation in breast cancer, pathologists could segment the nuclei with image analysis algorithms without having to estimate in a subjective way.
Recommended article

Article • Digital pathology
Bridging the gap between pathologist and algorithm
Digital pathology can gain huge benefits from rapid image search and the effective extraction of knowledge from large medical archives via artificial intelligence (AI). It facilitates identification of anatomical and pathological similarities, significantly enhances the clinical workflow, and ultimately paves the way for more informed diagnosis and better patient outcome. Effective archive…
AI algorithms are also particularly helpful in immune infiltrate quantification, an important prognostic feature that proves more difficult to assess in colon cancer than in breast. ‘You have more tissues, muscle, fat, etc.,’ he said, ‘and you only need to count a number of infiltrating inflammatory cells in a tumour area, so all areas must be recognised,’ Finding metastases in breast cancer is a tedious task in which pathologists typically have to put under and look at 50 slides, a process that can take up to 20 minutes. The missed rate is high. Van Diest and colleagues, who recently revised the original material from a big breast study, found that pathologists missed 23% of central node pathologies. ‘We need something to do a better job. AI did get better results than very experienced pathologists at detecting lymph node metastasis in breast cancer,’ he pointed out.
The problem remains that, so far, very few AI algorithms have been implemented in clinical cases. ‘We are not there yet,’ van Diest concluded. ‘Workflow needs to be completely integrated and we need fully integrated image analysis, but we need to do this step by step.’
Profiles:
In 1996, Professor Paul J van Diest gained his medical doctorate followed by Board certification in Pathology at VU University medical centre (VUMC) in Amsterdam, where he was then Consultant Pathologist in the Pathology Department. In 1999 he became Associate Professor and, in 2001, full Professor. He moved to head the Pathology Department at the University Medical Centre (UMCU) in Utrecht in 2003. He is also Adjunct Professor of Oncology at the Sidney Kimmel Oncology Centre at Johns Hopkins, Baltimore, USA, and visiting professor at the University of Siena, in Italy. He is on the editorial board of 23 international journals, has published around 700 papers in peer-reviewed journals; personally supervised about 70 PhD theses, and been active as secretary and president of various international societies.
Lluís Donoso-Bach gained his medical degree from the School of Medicine of the Autonomous University of Barcelona in 1981. He completed his residency in radiology at the Hospital de Sant Pau in Barcelona, in 1992, and in that year was appointed Chairman of the Radiology Department at the UDIAT Diagnostic Centre. In 1998 he became its Executive Director. Since 2006 he has led the Diagnostic Imaging Department at the Hospital Clínic of Barcelona and been Professor of Radiology at the University of Barcelona.
17.09.2019