Digital breast tomosynthesis
The future of mammography
By Thomas Mertelmeier, Principal Scientist at Siemens AG Medical Solutions
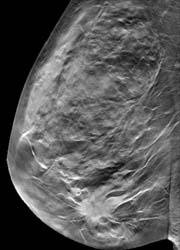
(Fig. 1b)
Full-field digital mammography (FFDM) offers many advantages over film/screen mammography. Whereas most commercial FFDM systems have shown to have superior physical image quality over their analogue counterparts, large scale clinical trials have demonstrated that FFDM seems equivalent to screen/film mammography with statistically significant diagnostic advantages for certain populations, such as women under 50 years old, women with dense breasts, and pre- or perimenopausal women.
The main limitation of projection mammography is not quantum or detector noise but the fact that the 3D anatomy is projected into a 2D image. Therefore, overlapping anatomical structures limit the radiologist’s ability to detect certain lesions. Digital tomosynthesis promises to overcome this limitation of projection mammography by reconstructing slice images.
The principle of breast tomosynthesis
Breast tomosynthesis is a 3D imaging technology that acquires 2D projection images of a compressed breast at multiple angles during a sweep of the X-ray tube. Objects at different heights in the breast are projected differently at different angles. The subsequent image reconstruction leads to a stack of slice images of the different depth layers parallel to the detector surface. The in-slice-resolution is predominantly determined by the detector resolution and usually much higher than the resolution between slices (‘depth resolution’) due to the incomplete sampling of the object within a relatively small angular scan range.
During the acquisition process the total dose is split among the single views. Because one voxel is probed by the same number of X-ray quanta as in projection mammography, a tomosynthesis scan needs approximately the same dose as a projection mammogram - under the assumption that the image detector does not excessively contribute to noise.
The objective of the Siemens tomosynthesis prototype device was to gain experience – together with our clinical partners – in how to provide a comprehensive solution for 3D mammography. The specific goals were to find the best acquisition mode for a tomosynthesis scan and to study reconstruction algorithms to optimise image quality. Another objective was to learn about the clinical performance and workflow of tomosynthesis.
The prototype is based on a Siemens MAMMOMAT NovationDR system modified for an X-ray tube motion over an arc of up to 50º. The detector used in this research system is a fast direct converting amorphous selenium detector, with an imaging area of 23.9 cm x 30.5 cm. The system is quantum noise limited, even for the lowest detector exposures used. A tungsten/rhodium anode/filter combination is used to keep the dose of one complete scan as low as that of one or two conventional 2D mammograms. The data of the examples presented here were acquired in a mode with 25 views and with scan time of 20 s.
Clinical benefits
Breast tomosynthesis has the potential to improve sensitivity in the detection of breast cancer due to reduced overlap of breast tissue, particularly in dense breasts. This may result in earlier cancer detection. Breast tomosynthesis may also lead to significant improvements in specificity as the 3D-analysis of the distribution of microcalcifications, and of shape and margin of lesions, might be easier. This could lead to a reduction in recall of patients and fewer biopsies.
Finally, digital breast tomosynthesis might eliminate the need for multiple exposures of the same breast. Thus it might lead to dose reduction, if only one tomosynthesis view, such as in the MLO orientation, is needed.
Image examples
At the University Hospital in Malmö, Sweden, human subjects are recruited under the direction of Dr Ingvar Andersson with informed consent in accordance with a protocol approved by the local ethics board. All human subjects also underwent standard digital mammography on a commercial FFDM unit.
One tomosynthesis example is shown in Figure 1. The 57-year-old woman with compressed breast thickness of 6 cm underwent tomosynthesis scans on each breast in MLO position. The mammogram of the right breast (Figure 1a) did not show the 2.8 cm palpable ductal cancer which is, however, clearly visible in the tomosynthesis data set, e.g. in the slice 2.5 cm above the patient table (Figure 1b).
A second example is shown in Figure 2. The breast of the 60-year-old human subject contains a well-marginated lesion (Fig. 2a) about 2.3 cm above the table and a microcalcification cluster in a different plane (Fig. 2b). The magnified view (insert) nicely demonstrates the detailed microcalcifications.
Conclusion - Currently, breast tomosynthesis is in research status, obtaining first clinical experience. The fundamental physical problems have been solved although many details need further investigation. One of the biggest challenges is related to data handling. For a tomosynthesis solution for routine clinical use, an efficient way of displaying, reading and archiving the huge amount of image data must be found.
Breast tomosynthesis has the potential to revolutionise mammography by significantly reducing the tissue overlap problem, inherent in projection mammography. This might lead to improved sensitivity and specificity, fewer recalls, fewer biopsies, less dose and less painful compressions. It can be expected that breast tomosynthesis will be used as a diagnostic tool in the beginning. However, after a learning curve and the diagnostic benefit have been proven, breast tomosynthesis would most likely be applied in the screening setting.
20.06.2007