Article • Entering a new age
AI predicts blood flow to the heart
Artificial Intelligence (AI) has, for the first time, measured blood flow to the heart to help predict which patients may suffer myocardial infarction or stroke. A research team at University College London and Barts Health NHS Trust and the National Institutes for Health (NIH) in the USA – are optimistic that AI analysis of perfusion maps will be a reliable, convenient and detailed new biomarker in cardiac patient care.
Report: Mark Nicholls
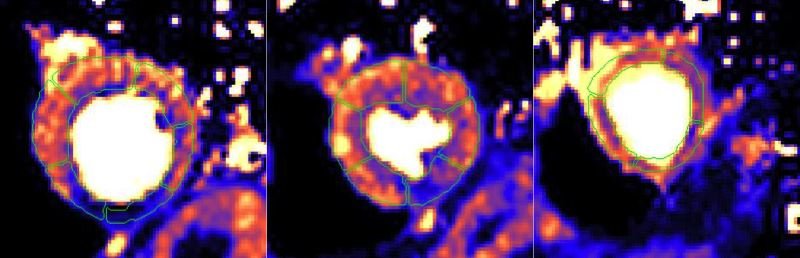
With myocardial perfusion reflecting the macro- and microvascular coronary circulation, the researchers explored the prognostic significance of stress myocardial blood flow (MBF) and myocardial perfusion reserve (MPR - the ratio of stress to rest MBF) in patients with suspected and known coronary artery disease referred clinically for perfusion assessment. Image analysis was conducted automatically using a novel AI approach and used to seek the associations of stress MBF and MPR with death and major adverse cardiovascular events, including myocardial infarction, stroke, heart failure, late revascularisation, and death.
A strong and independent predictor
‘In patients with known or suspected coronary artery disease,’ the study concluded, ‘reduced MBF and MPR measured automatically inline using AI quantification of cardiovascular magnetic resonance perfusion mapping provides a strong, independent predictor of adverse cardiovascular outcomes.’

Professor James Moon, who led the multi-centre research team, explained that measuring blood flow to the heart has been used for many years, but non-invasive blood flow assessments, including CMR (Cardiac Magnetic Resonance) imaging, remain difficult to analyse in a manner precise enough to deliver a prognosis or recommend treatment.
During the study, the UCL/Barts/NIH team made CMR ‘fully quantitative’ in measuring the actual amount of blood flow going to heart muscle, using AI to identify where the muscle is and making the process of working out where the blood pool is easier and quicker. From routine CMR scans from more than 1,000 patients attending St Bartholomew’s Hospital and the Royal Free Hospital the new automated AI technique – devised by Dr Peter Kellman and Dr Hui Xue at NIH – was used to analyse the images, enabling them to precisely and instantly quantify the blood flow to the heart muscle. By comparing the AI-generated blood flow results with the health outcomes of each patient, the team found that those with reduced blood flow were more likely to have adverse health outcomes.
By just measuring the large arteries, we are missing a chunk of the story about how our hearts are over our lifetimes
James Moon
Moon, Professor of Cardiology at UCL and the Clinical Director of Imaging at Barts Heart Centre (BHC), said that AI also could show findings the eye may not have previously seen. In addition to measuring blood flow to the heart muscle to highlight abnormalities and predict adverse outcomes, AI also identified what may be happening in a wider context with the patient, such as atherosclerosis and age-related stiffening of arteries. ‘By just measuring the large arteries, we are missing a chunk of the story about how our hearts are over our lifetimes, and that the microvasculature is clearly changing with age and diabetes and other parameters probably associated with chest pain and outcome. We think those are tractable with a new generation of treatments for the microvasculature disease function,’ Moon added.
As a new marker of disease, he acknowledges there is still some way to go to fully understand and realise the potential of this AI advance but also sees an opportunity to develop new therapies for patients with chest pain where the coronary arteries do not appear abnormal. However, the researchers believe their study has demonstrated the growing potential of AI-assisted imaging technology to improve the detection of heart disease and will have more diverse applications in cardiac care ‘It’s an indication that we are entering a new era of AI,’ said Moon.
Profile:
James Moon is a Professor of Cardiology at UCL and the Clinical Director of Imaging at Barts Heart Centre. His research focuses on developing mapping techniques with CMR and measuring diffuse myocardial fibrosis. Having published more than 300 papers, he is also looking at making scans faster and cheaper in improving quality as CMR expands.
15.09.2020





