Trends in image-guided therapy
Cardiac-CT for guidance of electrophysiologic interventions in patients with cardiac arrhythmias
For the third in his series of articles for European Hospital, Professor Stefan Schönberg of the Institute of Clinical Radiology and Nuclear Medicine (IKRN), University Hospital Mannheim, Medical Faculty of Mannheim, University of Heidelberg, invited colleagues at the Faculty's Cardiology and Radiology and Nuclear Medicine departments for a round-table discussion on:
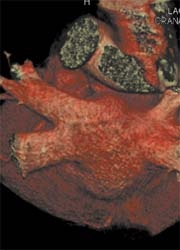
The pre-interventional identification of the middle lobe vein has direct implications on the therapeutic success, as it is often difficult to identify this small calibre vein at fluoroscopy.
A, B: 3-D volume-rendering reconstruction with different posterior views. C. Virtual angioscopy of the left atrium, view from left side (UL=Vein of upper lobe, LL=vein of lower lobe).
Over the last decades invasive cardiac electrophysiologic studies gained widespread acceptance for the diagnosis and treatment of cardiac arrhythmias. The spectrum of supraventricular and ventricular tachyarrhythmias that can be cured by catheter ablation increased significantly. More complex tachyarrhythmias such as atrial fibrillation (AF) or atrial tachyarrhythmias, premature ventricular contractions (PVC) or ventricular tachycardias in patients with complex congenital heart disease can nowadays be successfully approached.
Electrophysiological studies and ablation procedures are currently performed with manually deflectable catheters under fluoroscopic guidance. The fluoroscopic silhouette of the heart, together with excursions of the catheter during manipulation as assessed by different X-ray angulations as well as the electrical signals recorded by the distal electrode of the electrophysiologic catheter, are the tools to localise the target region for an effective therapy. Some major disadvantages are obvious. Frequent fluoroscopy is needed to control catheter movement which may lead to substantial radiation exposure time for both, the patient and physician. The cardiac anatomy is not visible during conventional procedures. Therefore, electrical signals recorded from the endocardium by the mapping catheter cannot be directly associated to the individual anatomy in case of conventional fluoroscopy.
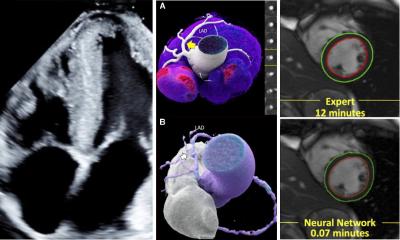
However, the last decade has witnessed the development of high-resolution non-fluoroscopic mapping systems and integration of CT or MRI data sets of cardiac anatomy. The three dimensional pattern of electrical activation during the arrhythmia, such as recurrent PVC or regular supraventricular or ventricular tachyarrhythmias can be reconstructed with sequential registration of the electrical activity from different anatomic regions of the heart. Furthermore, continuous non-fluoroscopic control of the tip of the visualised mapping catheter during manipulation in the heart chambers is possible. Thus, movement of the catheter can be performed with less or even without fluoroscopic control. Finally, the new generation electro-anatomical mapping systems offer the possibility to integrate high resolution cardiac CT or MRI images in the electrical map of the heart. Prior to the procedure a contrast-enhanced ECG-gated CT of the heart is performed. The cardiac chamber of interest is then reconstructed using 3-D postprocessing algorithms. Therefore, anatomical variations or abnormalities, which may have a significant impact on the outcome of the ablation procedure, e.g. variations of the pulmonary veins in patients with AF, can be detected and visualised prior to ablation (Figure 1). After acquisition of an electro-anatomical map during the invasive procedure the data sets can be merged to visualise both electrical and specific anatomic data (Figure 2). However, morphological information with a preprocedural approach has the disadvantage that it does not account for dynamic changes in anatomical structures and diameters.
Most recent technological developments enable the physician to steer catheters remote either by magnetic navigation or by robotic navigation with a remotely controlled flexible guiding sheath. Highly accurate and reproducible catheter placement and navigation is mandatory for successful and sustained ablation of the target. The knowledge of the individual and often significantly variable anatomy of the patient is essential for a safe and effective therapy. Thus, improved integration of anatomic CT or MRI data of the individual patient during electrophysiological procedures offers the opportunity to increase orientation, safety and efficacy in ablation therapy and to reduce radiation.
01.07.2008