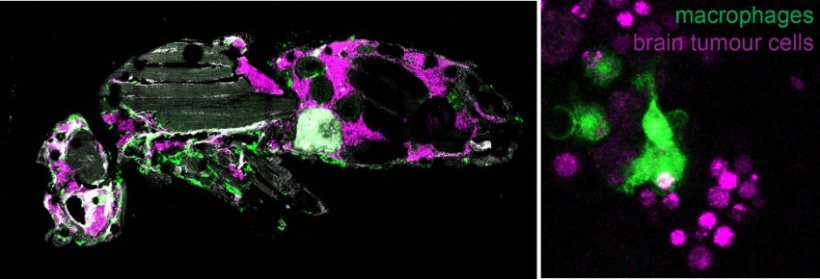
Right: Macrophages (green) engulfing and breaking down brain tumour cells(magenta).
Source: FORTH
News • Brain cancer reseach
Researchers reveal mechanisms that make tumours malignant
A new research study at the Institute of Molecular Biology and Biotechnology (IMBB) of the Foundation for Research and Technology-Hellas (FORTH) reveals how cancer cell intrinsic mechanisms and interactions with the microenvironment shape brain tumour growth and progression to malignancy.
The researchers published their findings in the international peer reviewed science journal PNAS.
Every year, 6 out of 100,000 people (children and adults) are diagnosed with brain, and other nervous system, cancer. Most of these patients will undergo therapeutic strategies combining surgery with radio- and chemo-therapy. However, only 33% of these patients will survive for more than 5 years. The life expectancy for the majority will be circa 18 months to two years.
Although there is a great research interest in neural tumours, current therapeutic strategies still remain ineffective. One of the reasons why brain tumours are hard to cure is because they are comprised of diverse cell types, both cancerous and normal. Only a subpopulation of these cells carry the combination of mutations that contributes to tumour emergence and maintenance. These cells are called cancer stem cells and it is thought that, to achieve tumour growth, they hijack developmental programs that, under physiological conditions, neural stem cells use to generate diverse cells types of neurons and glia during development and upon tissue repair.

Image source: FORTH
The scientific community believes that brain tumour progression to a malignant state is the result of an intricate interplay between deregulated intrinsic mechanisms in some Cancer Stem Cells and interactions with the tumour microenvironment, including cells of the immune system that infiltrate tumour masses.
In order to study mechanisms of tumourigenesis and malignancy in the brain, IMBB researchers Dr. Chrysanthi Voutyraki, Dr. Evanthia Zacharioudaki and Dr. Christos Delidakis, (Professor at the Biology Department of the University of Crete), used the fruitfly Drosophila melanogaster where, via a simple genetic manipulation, they generated a neural stem cell derived tumour.
They discovered that, as tumours progress to malignancy, developmental processes related to neuronal differentiation are shut down, whereas processes related to growth, metabolism and immunity are upregulated. By using sophisticated genetic tools, they showed that two pathways related to growth, more specifically the oncogene Myc and the Insulin receptor are necessary for tumour progression to malignancy. They also observed that macrophages from the host animals (the frontline soldiers of the body in the defense against invading pathogens) infiltrate tumours and resist/impede tumour growth inside their host. Live imaging revealed that macrophages engulf and consume tumour cells (a process known as phagocytosis) thus delaying tumour growth. When macrophages lack surface receptors involved in the phagocytosis of pathogens (like NimC1), they can no longer trap and engulf tumour cells thus tumours grow faster and kill their host earlier. Finally, while phagocytosis resists tumour expansion, the researchers discovered that macrophages also promote tumour growth by their natural propensity to produce reactive oxygen species (ROS) which contributes to faster host demise.
These findings contribute to a better understanding of neural tumour growth strategies and their complex interactions with their microenvironment. In the future, these Insights could serve as pointers for routes to follow in studies using mammalian systems. In the long run, they could enable the design and development of novel and effective chemical or immunological approaches to target specifically the proliferation of Cancer Stem Cells in brain related tumours (e.g glioblastoma) thus improving the life expectancy of brain cancer patients.
The research team at IMBB has received funding from the Hellenic Foundation for Research and Innovation (HFRI), Worldwide Cancer Research and Fondation Santé.
Source: Foundation for Research and Technology-Hellas
15.08.2023