Image cedit: Lloyd Mann
News • Covid-19 protection
Needle-free coronavirus vaccine enters clinical trial
Safety trials are underway for a Cambridge-led vaccine that could be used as a booster targeting Covid-19 virus variants and relatives that threaten future coronavirus pandemics.
The first volunteer is expected to receive the vaccine on December 14th at the NIHR Southampton Clinical Research Facility.

Image credit: Nick Saffell
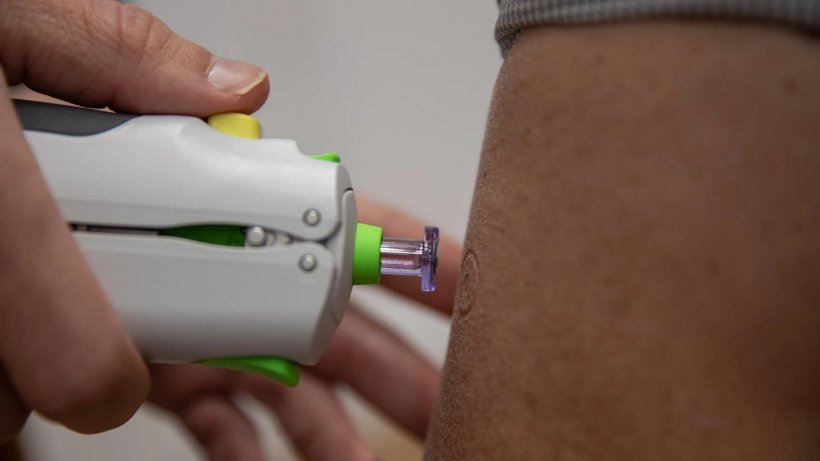
Developed by Professor Jonathan Heeney at the University of Cambridge and spin-out company DIOSynVax, this is a next generation coronavirus vaccine administered through a needle-free ‘injection’ – a blast of air that delivers it into the skin. This offers a possible future alternative to people who fear needle-based jabs. If successful, it could be scaled up and manufactured as a powder to boost global vaccination efforts, particularly in low- and middle-income countries. Professor Heeney said: “The response of the scientific and medical communities to the development and delivery of Covid-19 vaccines has been incredible, but as new variants emerge and immunity begins to wane we need newer technologies. It’s vital that we continue to develop new generation vaccine candidates ready to help keep us safe from the next virus threats. Our vaccine is innovative, both in terms of the way it primes the immune system to respond with a broader protective response to coronaviruses, and how it is delivered. Crucially, it is the first step towards a universal coronavirus vaccine we are developing, protecting us not just from Covid-19 variants but from future coronaviruses.”
How it works
SARS-CoV-2 uses ‘spike’ proteins on its surface to gain entry to host cells. These proteins bind to ACE2, a protein receptor on the surface of cells in our airways, allowing the virus to release its genetic material into the host cell. The virus hijacks the host cell’s machinery to allow itself to replicate and spread. Vaccines inform our bodies about what dangerous infections look like and how to respond to them. This is much safer than becoming infected with the live virus, because it avoids the life-threatening effects the whole virus can have. Immunisation arms our immune system to look out for and block virus, or destroy cells that carry the spike protein, protecting us from Covid-19 disease. Unfortunately, SARS-CoV-2 is constantly mutating and the virus spike protein itself is changing. This raises the prospect of ‘vaccine escape’, where changes to the spike protein mean the immune system is no longer able to recognise it.
This isn’t simply ‘yet another’ coronavirus vaccine as it has both Covid-19 variants and future coronaviruses in its sights. This technology could give wide-ranging protection to huge numbers of people worldwide
Saul Faust
To get around this problem, the Cambridge team searched for new types of antigens – key regions of the virus – that are the same across coronaviruses that occur in nature, including in animals that carry them, such as bats.
While most Covid-19 vaccines use the sequence of the RNA for the virus spike protein from the first isolated samples of the Covid-19 virus in January 2020, this new DIOSvax technology uses predictive methods to encode antigens like the spike protein that mimic the wider family of coronavirus antigens, thus giving wider protection. The body’s immune cells take up the vector, decode the DIOS-vaccine antigen and present the information to the immune system. This in turn produces neutralising antibodies, which block virus infection, and T-cells, which remove virus-infected cells. This technology is well-established and the vaccine plasmid DNA does not get taken up into human genetic material.
Professor Heeney added: “DIOS-CoVax vaccines target elements of the virus structure that are common to all known ‘beta-coronaviruses’ – those coronaviruses that are the greatest disease threats to humans. These are structures that are vitally important to the virus life cycle, which means we can be confident that they are unlikely to change in the future. These next generation DIOSvax vaccines should protect us against variants we’ve seen so far – alpha, beta, delta variants, for example – and hopefully future-proof us against emerging variants and potential coronavirus pandemics.”
The vaccine can be delivered pain-free without a needle into the skin, using the PharmaJet Tropis® intradermal Needle-free Injection System, which delivers the vaccine in less than a tenth of a second by spring-powered jet injection.
Volunteers wanted
The first trials of DIOS-CoVax are being delivered by the NIHR Southampton Clinical Research Facility (CRF). The trial team are calling for healthy volunteers aged between 18 and 50 in the Southampton area. Participants must have had both doses of a Covid-19 vaccine, but not their booster.
Professor Saul Faust, Clinical Chief Investigator and Director of the NIHR Southampton CRF, said: “This isn’t simply ‘yet another’ coronavirus vaccine as it has both Covid-19 variants and future coronaviruses in its sights. This technology could give wide-ranging protection to huge numbers of people worldwide. The people of Southampton and Hampshire have stepped up time and again to help find the vaccines that have unlocked the pandemic. We’re asking for their help again in developing this potentially game-changing vaccine.”
The phase I vaccine trial in Southampton will follow up volunteers for approximately 12 months to ensure it is safe. Payment will be provided for time and travel across 11 visits. Contact UHS.recruitmentCRF@nhs.net for further details.
Source: © University of Cambridge (CC BY 4.0)
14.12.2021