CT and US co-registration
RVS raises radiofrequency ablation success
Dr Alice Gillams, who heads the image guided tumour ablation programme at University College London Hospitals (UCLH), reported at this year’s ECR that Hitachi’s Real-time Virtual Sonography (HI RVS) fusion imaging technique can achieve more precise radiofrequency ablation (RFA).
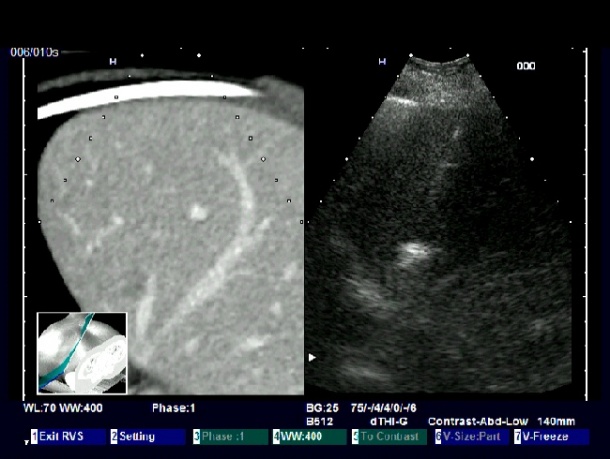

During the procedure a real-time ultrasound image is displayed simultaneously with a corresponding CT virtual multi-planar view, reconstructed from a stored volume data set. Thus the two imaging modalities provide a direct comparison of lesion location and more precise monitoring of interventional procedures -- without additional radiation exposure. The system is also time and cost effective.
RFAs have been performed at UCLH since 1994; the special co-registration software was installed in 2005. Thus, considerable experience has been gained over the past five years in the use of RVS, allowing progress in ablation for many different types of tumour in different organs, such as the liver and kidney, and occasionally, Dr Gillams pointed out during our EH interview, other ‘unusual’ sites.
The success of RFA strongly depends on the precise localisation and diagnosis of the extent of a tumour. In addition to the fact that the location of a tumour in a CT imaging slice varies according to patient position and respiration, the optimal depiction of several liver tumours depends on imaging at a specific point in the contrast enhancement cycle, which is often transient. Dr Gillams work has focused on minimising the problem by co-registration of the CT data set with real time virtual sonography.
‘Real-time targeting is essential because liver motion is not just translational but often rotational,’ Dr Gillams explained. ‘Knowledge of the exact location of a small tumour on CT often allows us to appreciate subtle alterations in echogenicity on the ultrasound image, which can then be targeted. Once the two data sets are co-registered, the tumour can be located on the CT image and then targeted in real-time with ultrasound.’
Working with co-registration has shown positive results. ‘The way co-registering makes a difference is that we can perform the procedure much more precisely and much quicker, without the need for repetitive interval scans. So there are clear advantages regarding time and radiation dose.’
Of course there are tumours that cannot be shown with ultrasound, for example those in the hidden liver area, where the lungs descend in the costophrenic recess. The same is true for large patients or those with a fatty liver where the portion of the superficially visible liver attenuates the ultrasound beam. ‘Looking back at an ablation experience of more than 15 years we definitely can say that since we opted for the real-time co-registration package and the software, we could perform much more precise treatments in a lot of patients – so the patient benefit increased significantly. That has been recognised by a lot of hospitals starting ablation programmes and many of them are ready for this specific expenditure. So the method surely will be more widespread in the near future.’
The technology is mostly co-registered with CT, however, Dr Gillams added: ‘Every now and again, we co-register with MRI data sets.’
Co-registration advantages
‘Very often, you cannot see the tumours so well with just the ultrasound, or you may be able to see them but only transiently. So, if you can co-register them with another imaging modality, where you can see the tumours very well, then you can have the best depiction of where the tumour is and then, equally, the real-time feedback on your needle being inserted and positioned correctly within the tumour. So you have the best of both worlds.’
Asked why UCLH chose to use the Hitachi’s RVS, Dr Gillams pointed out that not many manufacturers produce the real-time co-registration package and special software. ‘You have some hardware, which is the electromagnetic sensor, but most of the development is the software. It’s something additional that you buy when you buy the Hitachi machine. I also find that the Hitachi has very nice interventional ultrasound probes, which allow easy intercostal access and you can even insert your needle through the middle of one of their interventional probes. ’
07.07.2010






