News • Combined against corona
COVID-19: promising drug combination opens up new therapeutic avenues
As the COVID-19 pandemic continues, finding a treatment to effectively fight the disease remains a major research challenge.
Researchers from Inserm, CNRS, Université Claude Bernard Lyon 1 and ENS Lyon within the International Center for Infectious Disease Research (CIRI) have developed a unique strategy for selection, evaluation and repositioning of drugs already on the market to assess their effect against SARS-CoV-2. They have also developed several very relevant preclinical models of infections, using human respiratory epithelia of nasal and bronchial origin reconstituted in vitro. The researchers demonstrate that the association between the molecule remdesivir, used in the treatment of Ebola, and the antihypertensive agent diltiazem could provide a significant benefit in patients with COVID-19.
The scientists published their results in Cell Reports Medicine.
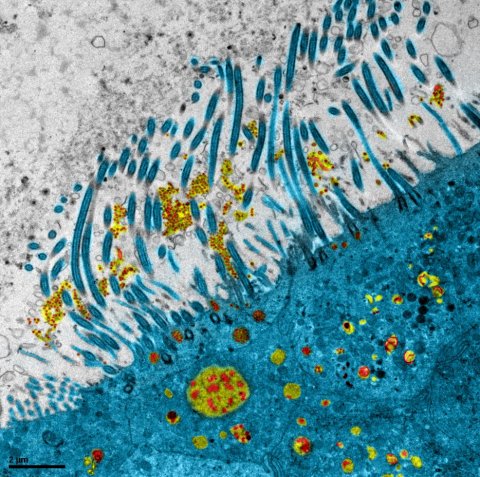
© Manuel Rosa-Calatrava, Inserm; Olivier Terrier CNRS; Andrés Pizzorno Signia Therapeutics; Elisabeth Errazuriz-Cerda UCBL1 CIQLE. VirPath. Colorized by Noa Rosa C
As part of the REACTing program coordinated by Inserm, the VirPath team led by Inserm researcher Manuel Rosa-Calatrava at the International Research Center for Infectious Diseases (Inserm/CNRS/Université Claude Bernard Lyon 1/ENS Lyon) is working on repurposing existing drugs for new therapeutic indications in viral infections.
To test the therapeutic efficacy of these molecules against COVID-19, the team began developing and characterizing experimental models of viral infection in February. This involved the in vitro reconstitution – as close as possible to human physiology – of human respiratory epithelia of nasal and bronchial origin. “We have been using these preclinical infection models, which are highly predictive of in vivo infection, for several years,” clarifies Rosa-Calatrava. The researchers have also developed protocols for viral-genome and infectious-particle quantification. Their observations and analyses confirm and supplement current knowledge of the mechanisms of SARS-CoV-2 infection and virus-host interactions. “One particular observation from our models infected with the virus was the induced production of interleukin IL6, which is a marker of the disease’s severity,” says Rosa-Calatrava.
Diltiazem is an antihypertensive used in the treatment of angina pectoris. It has already been characterized and repurposed by VirPath researchers to strongly stimulate the endogenous antiviral innate immune response, particularly against influenza- and respiratory viruses. The human toxicity of these two repurposed molecules has also already been evaluated, significantly reducing the time needed for their clinical development in the new SARS-CoV-2 indication.
The results of this study show a significant reduction in viral load in SARS-CoV-2 infected epithelia when treated with remdesivir. This effect is increased when diltiazem is added in combination. “By stimulating the innate immune response of the epithelia, diltiazem potentiates the effect of remdesivir and makes it possible to reduce doses. This bearing in mind that remdesivir presents some in vivo toxicity in addition to being a very costly drug,” emphasizes Rosa-Calatrava.
The team is continuing its preclinical tests with this dual therapy in animal models and hopes to launch a clinical trial as early as next winter if the positive results are confirmed.
Source: Institut national de la santé et de la recherche médicale (Inserm)
28.07.2020