Classifying COPD
Multi-detector CT scanners acquiring high-res 3-D data volumes offer new possibilities
Many smokers continue to use tobacco despite high taxes and drastic health warnings. Indeed, in industrialised countries, tobacco-related mortality rates or illnesses are increasing. These include chronic obstructive pulmonary disease (COPD), the fourth leading cause of death in adults – and rising.
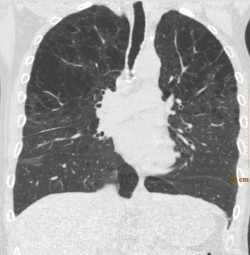
emphysema
COPD has many faces, so treatment needs to be tailored to the patient’s individual health status. Today, multi-detector CT scanners that can acquire highres 3-D data volumes offer new possibilities to differentiate,
phenotype and quantify COPD.
Dr Cornelia Schaefer-Prokop, specialist physician at the Radiology Institute of Meander Medical Centre in Amersfoort, the Netherlands, is an expert on COPD classification. ‘Typically it is a combination of emphysema and chronic bronchitis. The diagnostic workup includes clinical pulmonary function tests and the severity of airflow limitation is classified from one to four on the GOLD scale – the Global Initiative for Obstructive Lung Disease.
‘However, spirometry assesses only the function of the overall respiratory system; it does not provide any information on either the severity or the dominance of the individual components of COPD, namely emphysema and bronchopathy. This is unfortunate, since recent research has shown that precisely this information is crucial for a more targeted therapy.’
Both pathologies have characteristic features: emphysema is the enlargement of certain air spaces that causes the destruction of the alveolar walls, particularly as well as genetic factors are taken into account.
Computed tomography is excellently suited to differentiate emphysema and bronchopathy morphologically and, indirectly, also functionally, and to show the regional distribution of the pathologies. ‘Efforts are under way to develop processes for the quantitative assessment of the pathophysiological components,’ says Prof. Schaefer-Prokop, is in a research team of non-radiologists at the University of Nijmegen to draft quantification criteria. Density, she explains, is a particularly important parameter.
‘Where emphysema is concerned, the most frequently used threshold is -950 Hounsfield units. Everything below this value is emphysema. To gain more information on the bronchitis a CT scan is acquired of both
inspiration and expiration and the density values are compared. Expiration imaging shows that oxygen reaches the lungs but is not exhaled properly. This phenomenon called air trapping can be correlated to bronchitis or bronchiolitis, the obstruction of small airways.’
This type of differential diagnosis requires special software programmes, some of which are commercially available, while others are prototypes still being developed. ‘The problem is that these programmes, by either commercial providers or researchers, differ widely so that comparison is close to impossible’, Schaefer-Prokop explains. COPD phenotyping is still being evaluated and to some extent yields contradicting results.
For this new approach to be introduced in clinical practice, uniform standards have to be developed and a number of basic issues need to be answered such as: What exactly is being measured? How can the morphological values be correlated to the pulmonary function test results? Which factors can influence the measurements? ‘There are many patient and technology relevant details that must be taken into account’, the professor points out. ‘For example, the type of scanner, patient’s age, the depth of inspiration, the threshold being applied to density measurement, or the way data are reconstructed.’
From a clinical aspect, further research is necessary. While it is very likely that COPD phenotyping will lead to more personalised, earlier and thus better therapies, clinical studies corroborating this assumption need to be conducted.
21.02.2012