Cardiac resynchronisation therapy
From guidelines to daily clinical practice: Cardiac resynchronisation therapy is increasingly important in heart failure treatment. The therapy is complex, the demands to medical technology high. Holger Zorn reports
More than 3.6 million new cases of heart failure (HF) occur in Europe every year. Almost 40% of those patients die within a year of their first hospital admission; only 25% of men and 38% of women are still alive five years after their diagnosis. Around 20-25% of patients suffer from ventricular dyssynchrony (Source: Pacing Plin Electrophysiol. 2003 Jan; 26 (1 Pt 2): 155-7) and could benefit from cardiac resynchronisation therapy (CRT).
In 1994, Serge Cazeau, cardiologist at Val d’Or Surgical Centre in St. Cloud, France, implanted the first four-chamber pacing device into a 54-year-old patient with severe congestive heart failure (Pacing Clin Electrophysiol. 1994 Nov;17 (11 Pt 2) : 1974-91). In 2005, CRT was included in the ESC guidelines. Meanwhile, every year in Europe over 30,000 devices are implanted.
The cause of mechanical dyssynchrony is a cardiac conduction abnormality, a left bundle block (LBBB) with delayed ventricular conduction, which leads to uncoordinated regional contraction/relaxation. Medication can only treat this indirectly.
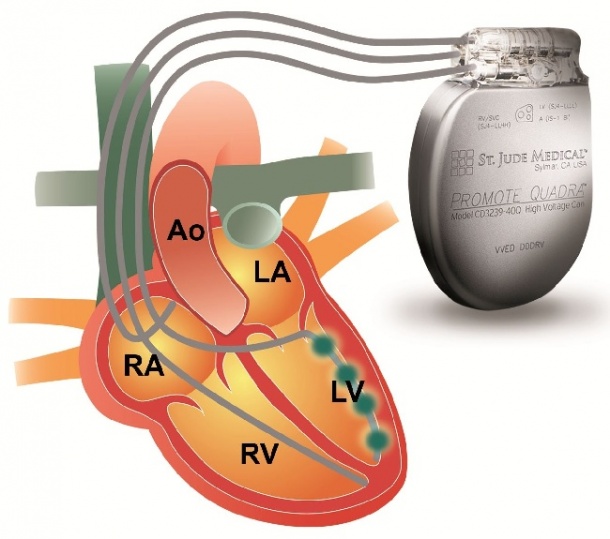
Cardiac resynchronisation therapy bypasses the blockade in the cardiac conduction system through the transvenous insertion of a third electrode (in addition to the normal pacemaker electrodes, which are inserted into the right atrium and the right ventricle). It is then positioned above the left ventricle via the coronary sinus in a coronary vein. Continuous stimulation of both ventricles forces them back into the right rhythm and the heart is resynchronised (see image).
14 randomised studies, involving 4,220 patients, proved that CRT can improve cardiac function and capacity in patients with severe HF. The hospitalisation rate decreased by 37%, mortality by 22% (JAMA2007;297(22) :2502-2514).
Newer studies led to an extension of its recommendation to mild HF patients and those with indication for a pacemaker: For instance, the MADIT-CRT study showed, for 1,820 patients from 110 centres in 14 countries, that in NYHA Class I and II patients with LBBB, early CRT intervention reduces the relative risk of all-cause mortality or first HF event by 57% when compared to ICD therapy (NEJM 2009;361 (14) :1329-38). Study leader Arthur Moss (University of Rochester) said: ‘CRT-D therapy offers the same advantages for ischaemic as well as non-ischaemic patients. The data of the MADIT-CRT study are convincing and contribute towards closing the gap in our understanding of heart failure class I and II.’
Around 5-10% of transvenously implanted CRT systems do not succeed. Special implantation aids are therefore as much the objective of continuous development as new electrodes. Left ventricular electrodes are now mostly not actively positioned with screws but via traction through the electrode shape and its surface friction against the vascular wall in the target vessel.
Frank Amberger, Senior Product Manager at St. Jude Medical GmbH, explains: ‘Because of this passive fixation the electrode can’t just be implanted in any given position; its position depends on the course of the veins in the heart.’ If transvenous access is not possible, epimyocardial electrodes offer an alternative. ‘These electrodes,’ he says, ‘are screwed in or surgically sewn into the wall of the left ventricle via mini-thoracotomy after opening of the pericardium.’ Non-surgical access, e.g. via subxiphoid puncture of the pericardium, are under development (Card Electrophysiol Clin 2010;2(1) :135-46).
However, an implanted CRT device does not always improve HF; only 35% and 22% respectively of patients are classed as responders and super-responders, defined by a reduction of left-ventricular end-systolic volume (LVESV) by 15-29% or 30% respectively and more than six months after the intervention. 21% of patients are non-responders, with an LVESV reduction of less than 14%, and 22% are negative responders, with LVESV actually increasing (JACC 2009;53:483-490) – for Dr Amberger enough potential for optimisation of CRT, be it through better patient selections, identification of the optimum location for stimulation, or through improved stimulation-algorithms.
Analysts also recognise this potential: In a 2008, a Frost & Sullivan study determined the market volume of CRT for seven key Western European regions -- Germany, France, Italy, Spain, the UK, Benelux and Scandinavia. Based on a figure of €832.5 million for 2008, F&S forecasts €1.943 billion for 2015, with a compound annual growth rate of 9.5%.
20.10.2011





