© Gerald Lip
Article • The iCAIRD project
AI to aid Scottish breast screening
Implementation of artificial intelligence into Scotland’s national breast screening service is moving closer following an initial success with a trial project.
Report: Mark Nicholls
While Scotland’s breast screening trial has delivered highs and lows, significant hurdles have been overcome in terms of approvals, governance and patient acceptance. The ongoing process of bringing AI into the mammography programme was outlined at the virtual Artificial Intelligence in Practice event organised by the British Institute of Radiology, in association with the Royal College of Radiologists.

In his presentation ‘Experiences with mammography AI evaluation, analysis and route to clinical practice in the iCAIRD project’, Dr Gerald Lip, Clinical Director of the North East of Scotland Breast Screening Service, outlined how AI is being lined up as an independent reader of mammograms in Scotland.
Lip, who is also the Chief Investigator of the Mammography AI project as part of iCAIRD (the Industrial Centre for Artificial Intelligence Research in Digital Diagnostics), explained that the process began in November 2018 with an Innovate UK grant, with contracts signed and data processing and information agreements set up as the partnership involving Kheiron Medical and Canon, the University of Aberdeen, National Screening Services in Scotland and NHS Grampian took shape. ‘Most of 2019-20 was spent on physical infrastructure, getting permissions, meetings, satisfying information governance and building a virtual workspace for anonymising 80,000 mammograms from over four years in the North East of Scotland,’ Lip explained.
Paperless screening
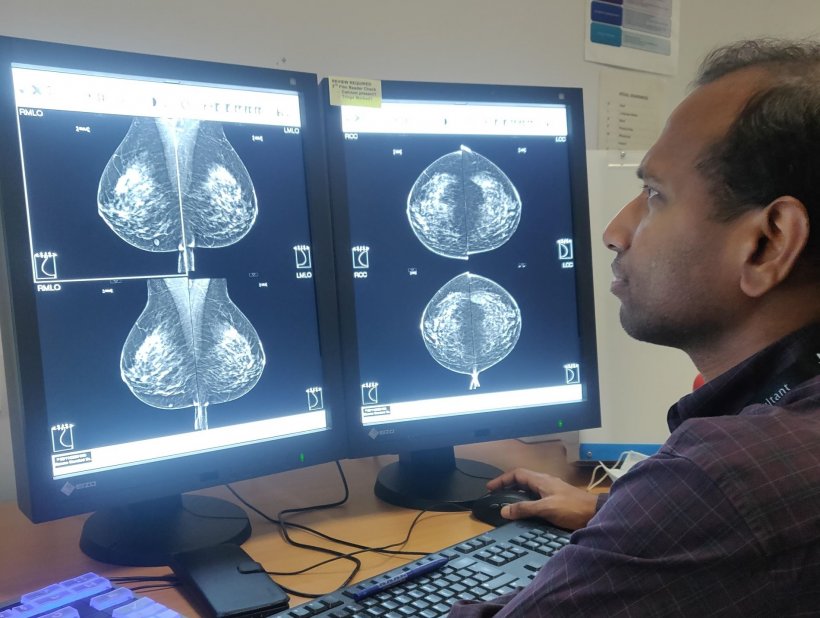
With the Scottish breast screening system already paperless, the data was readily available in a highly-structured data extract and transferred into the ‘safe haven’ for the project. Work progressed well in 2020 despite Covid, from Phase 1 of establishing feasibility and prototype, and into Phase 2 of development and clinical evaluation, clinical efficacy and safety data, with the project expecting to submit its first scientific papers this year.
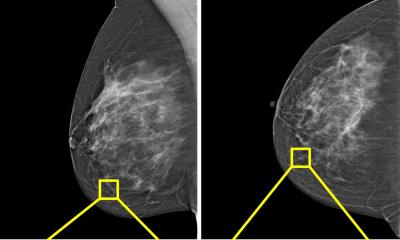
The project is now set to enter the Phase 3 stage of real-world testing, subject to research grants looking at the feasibility for implementation across multiple sites, regulatory approval, working through the health economics and subsequently into Phase 4 with initial health system adoption. ‘Phase 3 is the hard divide,’ Lip pointed out. ‘In a clean retrospective analysis in Phase 2 you can get good results but moving into clinical practice is a different story, especially in screening, because we are looking at evaluating this as an independent first or second reader.’ He also explained that the data used in the evaluation stage was not curated or enriched and covered four years with over 80,000 women, 240,000 images, and 700 cancers. As the project team knew what was there, they were keen to see what conclusions AI came to.
80-90% of women accept AI
Patient engagement has been important and positive with patient group surveys on women’s attitudes to AI in screening mammography. ‘In our population, 80-90% of women were very happy to have AI read their mammograms, particularly if a radiologist was involved,’ Lip reported. ‘Women who had an understanding of AI were more accepting of AI and if they could get results back faster, but safely, they were very happy with that process.’
Engagement with stakeholders such as the Scottish government and national screening services and research, advisory and governance bodies was a critical, yet time-consuming, step. ‘I’ve probably spoken to or corresponded with 40-50 different committees in the last two years, trying to put away misconceptions about AI. You find there are people in a different place to you and not all these groups were in perfect agreement,’ Lip reflected. ‘Covid was also an issue; there was a lot of prioritisation of Covid-based projects and suspension of non-Covid projects. We had ups and downs, but there were some highs: after six months of getting permission to move the anonymised data from 80,000 women into our safe haven, it was done over a weekend without affecting the network. Similarly, when we ran our first results trial for the AI project, it was 240,000 images in two days.’
The idea of AI, when it’s replacing a reader, is not to be the same as the average reader but to be as good as an experienced reader
Gerald Lip
The Scottish government has been supportive, as has NHSX, which has responsibility for setting national policy and developing best practice for NHS technology, digital and data. The project is now checking validation and clinical evaluation of the AI algorithm to ensure there is no bias as they look to moving into real-world practice.
Lip explained that, in breast screening, the usual cancer detection for a single reader is 6-8 cancers in 1,000 mammograms, whilst two radiologists combined may find 10-12 cancers. ‘The idea of AI, when it’s replacing a reader, is not to be the same as the average reader but to be as good as an experienced reader,’ he added. To move this forward into the Scottish breast screening system, the iCAIRD project team is designing a training module with AI integrated into it, that will read opinions and compare with expert readers. This is planned to be rolled out across six centres covering 20,000 mammograms a month to deliver ‘granular level of understanding’ of the data in the platform. The data training module, he explained, will act as a form of health technology assessment, rather than a formal randomised controlled trial, which will help the decision-makers and regulatory authorities to determine whether this can be adopted into the breast screening programme.
Questions remain about IT, data protection agreements, the use of the cloud and how screening readers get feedback on performance, but Lip is optimistic that, over coming months, the AI project will move closer towards implementation in the Scottish breast screening programme.
Profile:
Gerald Lip MD MSc is Clinical Director of the North East of Scotland Breast Screening Service, the Chief Investigator of the AI mammography project and an honorary senior clinical lecturer at the University of Aberdeen. He is a graduate of Trinity College Dublin where, along with his medical degree, he gained an MSc in Health Informatics.
04.03.2021