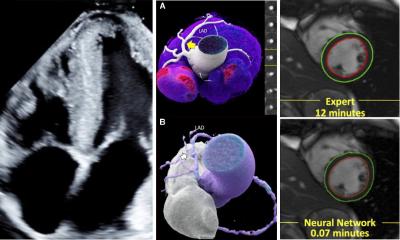
3D targeting of the heart
The heart is a structure in three dimensions and today we see it in three dimensions,’ Jose Luis Zamorano Gomez MD declared with satisfaction.
‘Even more, the heart is a volume in motion, contracting and relaxing, and the most significant development in cardiac imaging is that we can now study this motion in real time.’
‘In the past we needed to seek out experts in imaging technologies to quantify or measure accurately the morphology. Today, the imaging systems do this for us automatically, immediately capturing the volume, calculating the ejection fraction, quantifying the area of an annulus, measuring distances between structures and arteries.’
These quantification tools, he added, tremendously advance the reproducibility of results by greatly reducing inter- and intra-observer variability.
Prof. ‘Pepe’ Zamorano is currently leading the update for the European Society of Cardiology’s (ESC) recommendations for imaging inside the catheter laboratory with the European Association of Cardiovascular Imaging (EACVI). The working group is moving beyond a focus on transcatheter aortic valve replacement (TAVR) to cover a range of new procedures and new devices that have emerged since the recommendations were published in 2011.
This year, at the ESC congress in Barcelona, he will present his review of new imaging tools during the Spotlight Symposium on 1 September 2014, which aims to be devoted to ‘Innovation in Interventional Imaging.’
‘There are two innovations I will present that will have a high impact for interventional cardiology, and specifically for TAVR [transcatheter aortic valve replacement],’ he explained. ‘The first, a new 3D echography system, will show how interventions can be better planned and that the imaging information can go further to help guide the selection of the prosthesis.
‘Then, once we move to the cath lab, I will show how interventions can be guided in real time guidance with synchronised image fusion of fluoroscopy and 3D echo.
The cardiology centre at the Ramón y Cajal University Hospital in Madrid, headed by the professor, is the European reference centre for both systems.
Developed by Siemens, cutting edge planning tool for TAVR procedures generates off-line a 3D image of a patient’s native valve with an automatic generation of morphologi-
cal data that are very relevant for an interventional cardiologist, such as the area of the valve, the area of the annulus, or the distance to the coronary arteries, he pointed out.
The immediate application of this tool is a major advance to address the critical issue of valve implantation, yet the power of this technology also inspires new possibilities, he confirmed.
‘In a very near future, with all of this actual anatomical information and quantification, why not take it to a next step where we will be able to truly simulate the implantation of a specific valve in order to see what actually will happen, to see the positioning and the different aspects of the intervention in advance.’
Such modelling would become a predictive model for patient complications, identifying what is unique for each patient, and matching these conditions against a library of specific valve types and sizes. ‘This is where we hope to go with this technology and I have already spoken with the engineers who see no reason it can’t be done,’ the professor revealed.
The second focus for his presentation of innovation in interventional imaging is the EchoNavigator developed by Philips Healthcare.
The real-time fusion of 3D cardiac ultrasound with fluoroscopy is ‘something cardiologists have never seen before,’ he said.
In 99.9% of cardiac interventions, the interventionist is guided by conventional angiogram that requires interpretation of where the wire and prosthesis are located by watching a flat greyscale image that never actually shows the interventionist the heart.
By placing on the same interventional monitor a synchronised image of the heart, the use of 3D echo guidance becomes more intuitive.
‘With EchoNavigator we can now superimpose over the angiogram the anatomical structure at the same moment as the angiogram shows the wire for positioning the valve prosthesis. There is no longer any question of interpreting the image,’ Prof. Zamorano confirmed, ‘because we are seeing the anatomy exactly the way it is.’
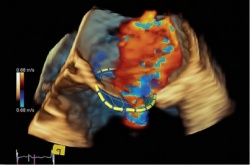
EchoNavigator use during left atrial appendage closure: fluoroscopy and 3D live-echocardiography fusion.
For left atrial appendage (LAA) closure, fluoroscopy for catheter and device visualisation and echocardiography in 2 and 3D for anatomy and soft tissue imaging are most frequently used. A new navigation system (EchoNavigator, Philips Healthcare, The Netherlands) has been introduced that allows synchronisation and fusion of echocardiography and fluoroscopy images in real time, as an aid during structural heart disease interventions. In the image on the left we can see the 3D transoesophageal echocardiography image of the LAA closure device during introduction into the LAA. The image on the right shows fusion between the 3D echocardiography and fluoroscopy images with the same orientation, during LAA closure device implant. It allows visualisation in one same screen, fused and in real time of both imaging techniques, as well as what will help during procedure guidance and device deployment. (Prof. Zamorano, Dr A González-Gómez. University Hospital Ramón y Cajal, Madrid, Spain).
A 3D echography data set is reconstructed with the cutting edge planning tool by Siemens Healthcare to show color Doppler flow across a reconstructed mitral valve (Courtesy of University Hospital Ramón y Cajal in Madrid).
Profile:
Professor Jose Luis Zamorano Gomez is Head of Cardiology at the University Hospital Ramón y Cajal in Madrid. A Fellow of the European Society of Cardiology (ESC,) he currently chairs the ESC Guidelines Committee and he is a past-President of the European Association of Echocardiography of the ESC. He is also on the editorial board of many leading journals, including the European Heart Journal and JACC Cardiovascular Imaging.
31.08.2014