Using light to control the cell
Researchers from the University of Heidelberg and the German Cancer Research Center (DKFZ) have developed a new method that uses light to control processes in living cells.
This system facilitates studies on the movement of proteins within cells and is of interest for both basic and applied research. The scientists have now published their results in the journal “Nature Communications.”
The new system is called, LINuS, like the companion of Charles Schultz’s cartoon dog Snoopy, and stands for “light-inducible nuclear localization signal.” In other words, it is a signal that can be used to lure proteins into the nucleus. The new method enables researchers to use light to interfere directly with processes in living cells. One advantage of the new method is that LINuS can be turned on and off like a light switch, and this can even be done several times in a row. In addition, it does not leave any other traces in the cell that might affect scientific observations. As opposed to regulation by chemical signals, LINuS also facilitates the investigation of individual cells in a unified cell structure. LINuS can be attached to any protein with the aid of a short DNA sequence so that it can be universally applied to the study of many types of processes in mammalian and yeast cells. Moreover, it is possible to vary the intensity and duration of the signal through light exposure. “We are able to ‘customize’ various versions of LINuS to the individual needs of the protein under investigation,” says study head Professor Roland Eils, a researcher at the German Cancer Research Center (Deutsches Krebsforschungszentrum, DKFZ) and the University of Heidelberg. “This opens up many new potential applications.”
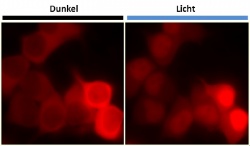
The new system is based on a light-sensitive protein in plants that is involved in their movement toward sunlight. “We have successively reengineered this plant protein into a light-sensitive protein shuttle service that works even in human cells,” says Dominik Niopek, first author of the publication. In the dark, the signal that mediates the transport of the respective protein into the nucleus is “hidden” in the LINuS appendix (the modified plant protein) and switched off. When exposed to light, it comes into the open and induces the transport of the labeled protein from the cell cytoplasm into the nucleus. If, for example, this protein is a transcription factor, it induces the activation of specific genes and thus directly impacts various functions of the cell. In a first step, the new system was tested using a fluorescent “reporter” that permitted observing the travel of the fluorescent protein directly under the microscope. The scientists also proved that LINuS can be used to interfere directly with basic cellular functions. Simply by turning on the light, they were able to switch on specific genes and even to induce cells to divide. In cancer cells, cell division often proceeds rapidly and runs out of control, resulting in genetic defects that promote tumor growth or resistance to specific drugs. Additionally, genetic repair mechanisms in cancer cells are frequently defective or even completely fail. “All these processes rely on a complex movement of signaling proteins, which is well-coordinated in healthy cells,” explains Barbara di Ventura, who leads the working group “Synthetic Biology” in Eils’ department. “Research on this is currently still in its infancy and can now be brought forward using LINuS.”
“Its key to future research is the possibility of spatial and temporal control of the activity of specific genes,” Dominik Niopek explains. “It is not enough to simply turn a protein on or off in a cancer cell. Within the cell, the movement of cancer-relevant proteins such as the guardian protein p53 is equally important. We can now use LINuS to study this.”
Roland Eils and Barbara Di Ventura have been working in synthetic biology for several years. This up-and-coming research field concerns the development of tools to equip cells with entirely new properties and thus to open up new applications in medicine, biotechnology, environmental technology, and other fields. LINuS is an important addition to researchers’ toolbox of methods. “The field of optogenetics, which uses light to control protein activity, is currently developing rapidly because light is an ideal, selective ‘switch’ that can be used to control proteins in individual cells with almost no side effects,” Barbara Di Ventura says. Last year, a student team from Heidelberg supervised by Eils, Di Ventura and Niopek won the Grand Prize in Synthetic Biology at the iGEM 2013 World Championship Jamboree in Cambridge, Massachusetts.
23.07.2014