Article • Organ donation
The liquid biopsy
Donor transplant rejection and cancer relapse have two things in common: early recognition is vital and monitoring is hugely challenged.
Much time and money invested in biomarker development for post-operative monitoring and early recognition of acute or chronic rejections has gained little success. Now a likely breakthrough lies in a test developed by Chronix Biomedical scientists along with Professor Michael Oellerich at Göttingen University Medical Faculty. The Oellerich team’s entirely new approach – using droplet digital PCR to measure cell-free DNA in transplant patients’ blood allows early recognition of rejection. ‘The great advantage of this new biomarker is that it yields direct information on the integrity of the organ,’ Prof. Oellerich explains.
The team recently received the American National Academy of Clinical Biochemists (NACB) prize for the research results presentation.
Immense technical challenges

‘The first challenge we faced was that circulating cell-free nucleic acids from the donor organ are diluted by the nucleic acids being released from all the other cells in the organ recipient’s body, recalls Julia Beck PhD, Senior Scientist/Lab Supervisor at Chronix Biomedical. ‘Therefore, we had to find some way of detecting a very weak signal from this organ against a very strong background signal from other DNA that stems from the recipient.’
The study leader indicated another obstacle: ‘We wanted to be in a position to rapidly detect the circulating cell-free nucleic acids produced by the donor organ – if possible, within a day – to facilitate timely intervention by the surgeon undertaking the treatment.’
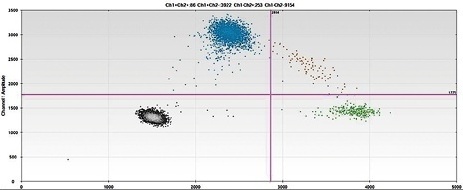
Due to weak signal strength standard PCR could not be used: ‘This method involves amplifying specific DNA sequences that contain a single nucleotide polymorphism - SNP. The organ and recipient must have different alleles at this location. The organ allele must then be detected against a huge background of alleles in the recipient,’ Dr Beck explains. Droplet digital PCR provides a solution: ‘This method no longer takes place in a single reaction vessel, but the sample is distributed across many tiny, individual reaction vessels in 20,000 “water-in-oil droplets”, each composed of one nanolitre of liquid. In essence, each molecule thus has its “own” reaction vessel.’

Each of these one-nanolitre droplets contains a template molecule – i.e. in other words, a target molecule, which means the relevant signal is not drowned out by background signals. Additionally, the droplets can be counted individually: after using allele-specific hydrolysis probes, droplets containing a template molecule produce a fluorescence signal after PCR. Dr Beck: ‘After marking the allele-specific probes with different fluorophores, a blue fluorescence signal is generated by the template molecule from the organ, while a green fluorescence signal is produced by the molecules that stem from the cells of the recipient.’
The donor organ releases DNA when cells die. ‘For example, at a specific point in time, we measure ten percent of alleles that stem from the organ relative to ninety percent of alleles from the cells of the recipient in the patient’s blood. This measurement shows us that a corresponding number of cells have died in the organ. If the signal increases from ten percent to thirty percent or more, then this is an indicator of the fact that too many cells are dying.’ At this point, the recipient’s immune system is attacking the organ, causing cell death.
Dr Schütz: ‘This method’s analytical simplicity is what makes it so attractive. We don’t have to qualify anything, don’t have to use circuitous routes for measurement through exclusion methods, rather more, we count, or the computer counts. In addition, it’s easy to convince any surgeon, as they can understand the method immediately. A huge advantage.’
A precondition: assays must be developed
The team had to develop a set of different PCR assays as a basis for the early warning system, i.e., combinations of probes and primers, because at least five polymorphisms, each with different alleles, should be available for each combination of recipient and organ. To this end, a set made up of 41 such positions was developed, which are then tested in advance in each patient. Each assay must be designed to discriminate between the two SNPs and produce the signal reliably. In an initial study conducted in collaboration with Prof. Otto Kollmar, Director of Transplant Surgery in Göttingen, patients were investigated after a liver transplant. This study, which confirmed the validity of the approach, is part of a multi-centre study on biomarkers in transplant medicine, funded by the German Federal Ministry of Education and Research (BMBF).
Long-term organ survival
Half of donor kidneys are still being lost within ten years, and this against a background of a serious shortage of organs
Michael Oellerich
Based on changes in data collected during monitoring, the surgeon receives information on whether intervention is required and the immune system medicinally suppressed, or whether a biopsy must be taken for further tests. Prof. Oellerich: ‘Half of donor kidneys, for example, are still being lost within ten years, and this against a background of a serious shortage of organs. A high percentage of patients exhibit levels of immunosuppression that are too low under standard therapy, resulting in chronic rejection in the long term; or, immunosuppression is too high, with resultant cardiovascular side effects, opportunistic infections, malignant diseases and kidney damage. For the first time, the circulating cell-free DNA gives us the opportunity to directly investigate donor organ integrity in a highly specific manner.’ This method, he adds, can be combined with conventional drug monitoring and thus be used to achieve fine adjustments to immunosuppression for kidney and other organ transplants. Prof. Oellerich: ‘The optimised treatment thus achieved for such patients could result in relevant improvements in long-term survival of the transplants.’
Detecting the genetic differences
Droplet digital PCR, Dr Schütz points out, ‘also has applications in therapeutic follow-up after the resection of benign and malignant tumours and over the course of monitoring. To date, when a local relapse or metastases had developed in patients, this could only be detected using costly and complex techniques such as CT or MRI.’ It is well-known that all malignant tumours differ genetically from the rest of the body. ‘Therefore, our approach was to seek out these genetic differences, preferably in the primary tumour after it had been surgically removed, or when a biopsy was available. We then attempt to detect these differences in the blood, using the same approach as we described for transplants. We can assume that cancerous tissue is growing again as soon as we detect a genome sequence from the degenerated cancerous tissue.’
This application is far more complex, says Dr Urnovitz. No cancerous cell is the same. ‘We must find a needle in the haystack.’ Based on which changed genome sequences in tumours can be identified, his firm developed and patented the method in late 2012.
Clinical acceptance
The Göttingen research team now aims for multi-centre studies to examine the new test’s application to heart and kidney transplants in a larger patient cohort.
Communicating these new approaches to clinicians is now the most important task: ‘We want to help patients at an early stage, avoid mid- and long-term damage and free the healthcare system from prohibitive costs. Our rapid, successful, low-cost method is well on the way to becoming the gold standard. To this end, we must communicate the method and, above all, the advantages to our colleagues.’
08.10.2014