Open for all patients
Hitachi Medical Systems UK has been awarded the contract for OASIS™ 1.2 Tesla high-field open MRI by InHealth MRI Centre at Mayday Hospital, Croydon – the high performance system with a truly open architecture available in the UK. This unique product combines unrivalled patient comfort together with first class imaging comparable to that of a closed bore system.
CT and MRI have become indispensible in radiological diagnostics. However, conventional MRI systems with a narrow, restricted scanning tunnel can cause great anxiety to the patient and with almost one in every four patients suffering from claustrophobia the examination either does not take place or can only go ahead if sedation is used. The OASIS™ combines a truly open design together with high field strength providing optimal image quality comparable to that of a closed bore system. The 270° angle of vision allows the patient an unobstructed view during the scan for a far more comfortable experience whilst simultaneously giving the clinician a clear view of the examination in progress.
The bariatric issue is a burgeoning problem in Europe demanding a radical rethink in MRI design whilst maintaining the necessary image quality. The OASIS™ provides the widest patient table, largest flex body coil and specific bariatric scanning protocols within an open MR environment meeting the bariatric imaging challenge and reducing rejection rate.
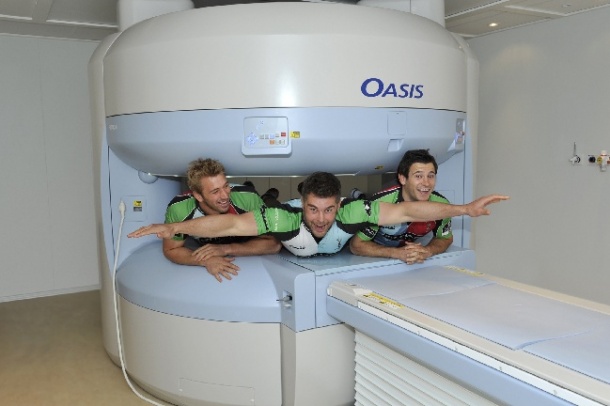
Mayday Hospital has officially reopened its fully renovated state-of-the-art MRI unit marking the occasion with a visit from three nationally renowned Harlequin rugby players who demonstrated the true openness of the system!
The design is unique and can accommodate patients who cannot fit into a conventional closed bore type magnet. Anxious or nervous patients are easily reassured and those suffering from claustrophobia don’t always have to be sedated. Referrals for patients suffering from claustrophobia to Mayday MRI from London and the South East has risen significantly since installation in April 2010 proving that there is an essential requirement for this technology.
Powerful, high performance MR electronics with fast gradients and multi-channel RF technology provide diagnostic performance comparable to that of a 1.5T MR but with the advantage of the open architecture design.
29.06.2010






