Extra-aortic counter pulsation
The C-Pulse, manufactured by Australian-American company Sunshine Heart, Inc. is neither a pulsatile artificial heart nor one of the well-known non-pulsatile left heart support systems
Picture: Dr. Nickolaus (DHZB)
Picture: Dr. Nickolaus (DHZB)
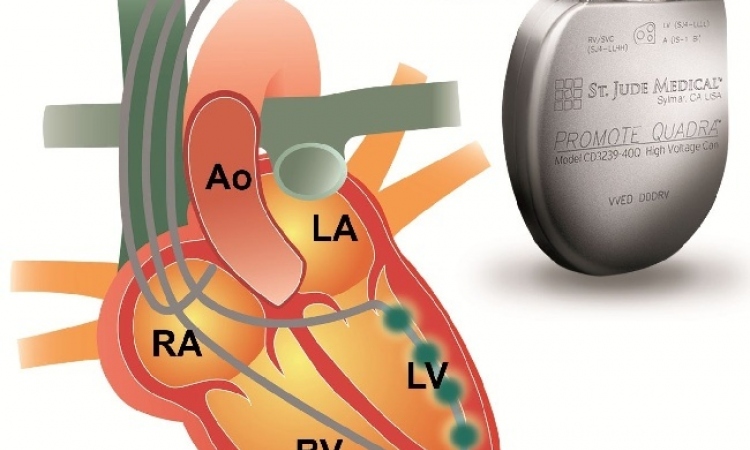
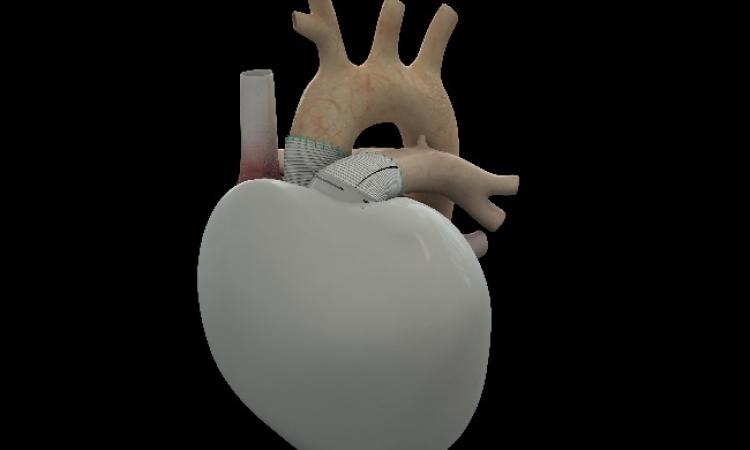
However, the device appears to be suitable to help slow down or even stop symptoms in patients with moderate chronic heart failure. The technology is innovative: the principle of counterpulsation (CP) is not applied intra-aortically but extra-aortically. A cuff is placed around the ascending aorta and triggered by an ECG-electrode attached to the left cardiac apex epicardially (image 1, 2). When the left ventricle contracts and the blood has been ejected into the aorta, the balloon inside the cuff is blown up and compresses the aorta slightly so that more blood is retained and flows into the coronary arteries, directly improving the heart’s oxygen supply. Draining the balloon just before each contraction of the ventricle temporarily also lowers resistance in the aorta and alleviates work for the heart.
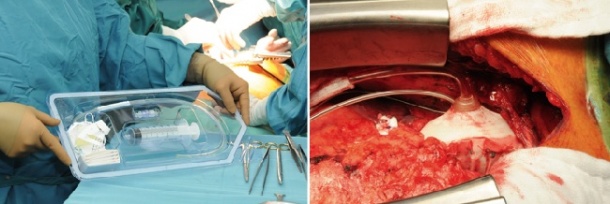
The device was first used in humans in May 2005 at Auckland City Hospital in New Zealand. Dr William S Peters, the system’s inventor, has been implanting the C-Pulse in Australia since 2010 (source: J Heart Lung Transplant. 2010;29:1427-32).In May 2013 he oversaw the first European implantation of the system at the German Heart Institute Berlin (DHZB). Professors Roland Hetzer and Thomas Krabatsch, of the DHZB, and Dr Holger Hotz, of the Cardio-Centrum Berlin, have now fitted three patients with the C-Pulse (image 3) who had not responded to prior cardiac resynchronisation therapy (CRT) in the context of a pan-European, multicentre study. Prof. Krabatsch sees two advantages: ‘The system does not come into contact with the patient’s blood circulation; therefore there is no need for permanent anticoagulation therapy. Moreover, the patient can turn the system off temporarily or even disconnect it – for example, to take a shower. Of course, this is not possible with other pumps that are fitted intra-aortically.’
The operation can be carried out without using a heart-lung machine and takes less time than the implantation of a classic blood pump whilst still being technologically complex. ‘The aorta has to be completely exposed so that it can be correctly covered by the cuff, Prof. Krabatsch pointed out, adding: ‘The ECG-electrode also has to be attached to the heart from the outside, most often at just the point that’s furthest away from surgical access, i.e. the sternotomy.’
Failing hearts in which drug treatment has been unsuccessful do not tolerate such manoeuvres haemodynamically without some restrictions. One patient required a Ventricular Assist Device (VAD) just a few days after the intervention.
These VADs are usually only implanted when patients are in a state of terminal heart failure, as bridge-to-transplant devices or destination therapy. But the C-Pulse has a different approach: The implantation at an early stage either at least delays or possibly completely removes the need for the implantation of a VAD. Dr Peter Göttel, Medical Director for Europe at Sunshine Heart, added: ‘The C-Pulse system bridges the gap between CRT-Non-Responders and the indication for an LVAD within the therapeutic range. It’s important that cardiologists who treat these types of patients mostly as out-patients are aware of the existence of new, less invasive methods of cardiac support such as extra-aortic counterpulsation. The C-Pulse alleviates patients’ symptoms with minimal impact on quality of life. In the future, we will also offer a fully implantable version. As the system does not require an implantable buffer battery, this technological advance will be possible soon.’
Studies involving 20 patients in Canada and the USA, and 50 patients in 11 hospitals across Europe, will now test the results gathered so far. These include, in Germany (alongside the DHZB), hospitals in Hannover, Duisburg, Düsseldorf and Erlangen in Britain a hospital in Glasgow, the Royal Brompton in London and Harefield Hospital, Middlesex; in Italy, hospitals in Milan, Padua and Turin.
Profile:
Professor Thomas Krabatsch gained his medical degree at the Humboldt-University of Berlin and trained as a cardiac surgeon at Berlin’s German Heart Institute. He has been Consultant for Thoracic & Vascular Surgery since 1999. In 2002 he wrote his habilitation on ‘Examinations of the clinical relevance and underlying mechanisms of transmyocardial laser revascularisation’
03.09.2013