Cardiac resynchronisation
This summer the world’s first implantations of Biotronik’s new ICD and CRT-D series (implantable cardioverter-defibrillators and cardiac resynchronisation therapy defibrillators) took place at the Spedali Civili Hospital, Brescia, Italy.
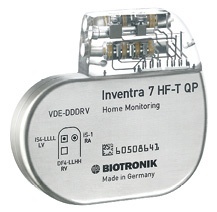
‘My ICD and heart failure patients are frequently indicated for MRI scans to diagnose potential comorbidities,’ said Dr Antonio Curnis. The researcher had implanted the firm’s ProMRI Inventra HF-T and Sentus quadripolar lead in a 73-year-old patient with congestive heart failure. ‘With the Biotronik devices, I know I can give my patients high-quality therapy and broad access to diagnostics,’ Curnis explained.
With Sentus quadripolar leads and the Inventra series, the manufacturer confirms that it is the first and only company to produce cardiac resynchronisation devices and leads for heart failure (HF) patients that are approved for MRI scans. As patients age, they may develop comorbidities, and MRI scans can be critical in diagnosing conditions such as stroke, brain tumours or orthopaedic conditions.
‘The quadripolar Sentus lead eases the implantation process by giving physicians access to challenging vessels. With CE approval in early July, Biotronik’s new implantable defibrillator series includes the industry’s first quadripolar left-ventricular leads to be approved for MRI use.’
In addition to ProMRI technology, the firm’s new ICDs and CRT-Ds reduce inappropriate shocks with MorphMatch morphology detection criteria and anti-tachycardia pacing (ATP) optimisation. ‘While delivering shocks at the right time can save patients’ lives, shocks should be minimised to appropriately control arrhythmias, improve patients’ quality of life and increase device longevity,’ Biotronik point out.
Dr Werner Jung, at the Schwarzwald-Baar Clinic, Villingen Schwenningen, Germany, successfully implanted a 72-year-old HF patient with a new ProMRI CRT-D from this bio-tech manufacturer. Speaking of the ‘exceptional quality of Biotronik products, the surgeon said: ‘Many heart failure patients are very ill and shocks put stress on the body and mind. By choosing a device with unique algorithms that reduce shocks, I can give my patients peace of mind and restore their sense of safety.’
The company has included its Closed Loop Stimulation (CLS) technology in ICDs for the first time. ‘CLS helps patients experience the most natural rate adoption possible by utilising their neurological information,’ the company explains. ‘It’s the only system that allows pacemakers, and now ICDs, to react naturally to patients’ physical as well as mental activity or stress.’
First to implant the new defibrillator series:
Antonio Curnis MD, Spedali Civili Hospital, Brescia, Italy
Werner Jung MD, Schwarzwald-Baar Clinic, Villingen Schwenningen, Germany
Tisiano Moccetti MD and Angelo Auricchio MD, Cardiac Centre Ticino, Lugano, Switzerland
Anthony Stanley MD, Sunninghill Netcare Hospital, Johannesburg, South Africa
Martin Winterhalter MD, SRH Clinic, Gera, Germany.
21.08.2014