Article • CEUS
Advancing contrast enhanced ultrasound
The ability to demonstrate blood perfusion as well as organ function using contrast agent enhanced ultrasound is quickly finding innovative uses in clinical practice.
Contrast-enhanced ultrasound (CEUS) has advanced rapidly since its first introduction. Today it is widely used as a primary imaging technique for a number of indications and pathologies. At a symposium organised by Bracco Imaging during the European Congress for Radiology 2017, the session chairman, Professor Jean-Michel Correas from the Necker University Hospital in Paris, France, introduced three practicing clinicians who demonstrated new applications for CEUS.
In 2016, an ultrasound arm of the Liver Imaging Reporting and Data System (LI-RADS) set forth criteria aimed at harmonising the interpretation of findings obtained at CEUS with those of traditional CT and MR criteria for hepatocellular carcinoma (HCC) lesions. According to Professor Fabio Piscaglia of the Medical and Surgical Sciences group at Italy’s Bologna University, while CEUS should not replace CT or MRI, the ability to integrate imaging modalities can be very useful for best patient management. He cited a situation in which neither CT nor MRI is conclusive as to whether detected nodules are benign or malignant. Here, CEUS proved invaluable in targeting specific nodules to select those that should undergo biopsy. The CEUS LI-RADS working group of the American College of Radiology adopted a conservative approach with stringent criteria to preserve an extremely high positive predictive value for the diagnosis of HCC and assure confidence that staging with ultrasound examinations can be trusted. Detected lesions are rated using the same scale applied to CT and MRI findings. Lesions classed as LR-2 to LR-3 have a low probability of being HCC, the probability of HCC is higher for LR-4 lesions and for those characterised as LR-5 there is a certainty of malignancy that virtually eliminates the risk of misdiagnosis.
Piscaglia presented results from a University of Bologna study where, across 300 nodules, a positive predictive value for LR-5 was found to be more than 99 percent for HCC. According to Piscaglia, inclusion of the LI-RADS classification of lesions in radiology reports can help to establish a definitive diagnosis of HCC and avoid the risk of a misdiagnosis with cholangiocellular carcinomas (CCC).

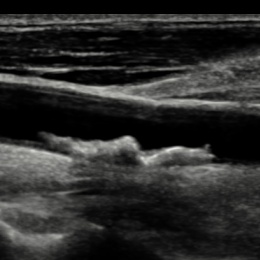
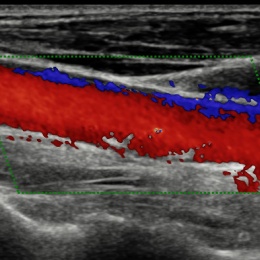
The standardisation of criteria also aids in communicating clear information to the referring hepatologist, not only to recommend biopsies, but also to assure optimal patient management in cases where the malignant lesion cannot be biopsied. The rating scale also helps in monitoring the progression of a specific lesion over time, even without a tumour size change. Supported and endorsed by ACR and European professional societies, LI-RADS is a dynamic document, Piscaglia said, continually expanding through refinements as new findings accrue and in response to clinicians’ feedback. Applying the exceptional vascular imaging properties of CEUS to the carotid artery, Professor Dirk-André Clevert, Section Chief of the Interdisciplinary UltrasoundCentre at the University of MunichGrosshadern Campus, demonstrated how these exams can aid in assessing a stroke risk and determining the best patient treatment regimen. An unstable carotid artery plaque causes approximately 25-50% of all strokes and about 80% of all strokes are ischemic, he said. ‘With reconstructed images from CT and MRI we can see the complete carotid artery and stenosis of the arteries. Yet, do we know the extent of the stenosis? And we need to think about the correct treatment for the patient, in light of the extent of the occlusion. A reliable ultrasound exam becomes essential in these unclear cases where a decision needs to be taken for stenting, for surgery, or for no treatment in the case of an occlusion.’

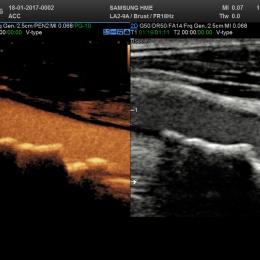
Following a preliminary scan using grey-scale B-mode, the next step for the radiologist is to turn on colour Doppler to assess the blood flow, Clevert explained. Yet Doppler ultrasound has some limitations and does not always provide full diagnostic differentiation of critical stenosis from complete occlusion. In a further step CEUS can provide more information to aid with the treatment of the patient with an ability to show the complete extent of the stenosis without artefacts. CEUS can also help assess stroke risk based on the contrast uptake inside the plaque, and thereby provide more conclusive information on the vulnerability (risk of rupture) of the plaque. Combined with the high temporal and spatial resolution of ultrasound, contrast enhanced ultrasound (CEUS) is well placed to study vascular diseases and improve the visualisation of pathologies, he concluded. At the Sichuan Provincial People’s Hospital, Chengdu, China, Dr Jun Luo has been exploring the usefulness of applying CEUS to the characterisation and BI-RADS classification of focal breast lesions. A particular feature of Asian women, he explained, is that they have relatively small and dense breasts, which can complicate interpretation of traditional mammography images. As a result, sonography is usually considered the primary clinical work-up tool in China.
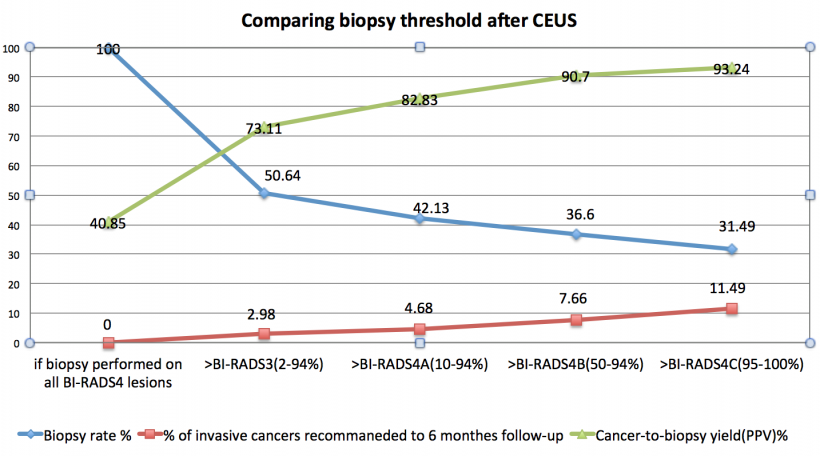
Japanese studies have similarly shown the effectiveness for ultrasound over mammography for Asian woman to detect early cancers, and a USA study demonstrated that findings with ultrasound are comparable to mammography. However, a limitation is a higher false positive rate using ultrasound with a resulting over-diagnosis rate ranging from five to as high as 50%. Due to this tendency for overdiagnosis, Luo stated provocatively, ‘BI-RADS is still not yet good enough.’ The key point in Luo’s analysis is that neither ultrasound nor mammography includes dynamic perfusion information that can be fundamental in the diagnosis of breast lesions. Additionally, without perfusion information, neither modality can assess the risk of malignant transformation of a benign lesion. The addition of perfusion information aids in clarifying whether suspicious masses are malignant, cysts or benign lesions. The potential with CEUS, he proposed, is to create predictive models that would maximise the reduction of false positives and minimise misdiagnosis of invasive breast cancers. Citing the results of a single-centre study that applied predictive models, the use of CEUS reduced the biopsy rate by up to 50% with only 2.98% missed invasive breast cancers that were detected only at the six month follow-up. The results have led to the start of a multicentre study in China to validate the findings. ‘BI-RADS can be improved by using contrast because it gives information on microvasculature, which is very important in differentiating benign breast nodules from malignant lesions,’ Luo said. ‘Contrast ultrasound may also predict the risk of malignant transformation among benign lesions if microvascular proliferation is observed, just as occurs in examinations of the liver.’
Profiles:
Professor Dirk-André Clevert is the Section Chief of the Interdisciplinary Ultrasound-Centre at the University of Munich-Grosshadern Campus, Germany.
Dr Jun Luo is the head of the Ultrasound Department at the Sichuan Provincial People’s Hospital in Chengdu, Sichuan, China.
Professor Fabio Piscaglia heads the Centre for Ultrasound of Internal Medicine at the University of Bologna, Italy.
27.11.2017