Key factor identified
Why is the immune system unable to combat HIV?
An international research group with essential participation of the Paul-Ehrlich-Institut, has identified NLRX1, a cellular factor of the human cell that is indispensable to the replication of Human Immunodeficiency Virus (HIV-1). This factor plays a key role in attenuating the innate immune system towards HIV-1. Until now, the significance of NLRX1 for the replication of HIV-1 and the attenuation of the immune system was not known. The novel research finding will lead to new therapeutic approaches.
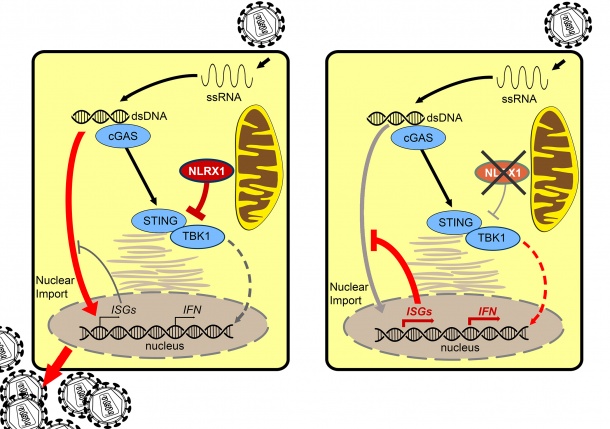
HIV-1 uses human proteins (host proteins) for its own replication and prevents the human immune system from combating it successfully. Dr Renate König, head of the research group “Cellular Aspects of Pathogen Host Interactions” of the Paul-Ehrlich-Institut, and her colleagues have been studying this phenomenon for a long time, asking why the human immune system fails to fight HIV-1. To identify the underlying reason could help develop both enhancers for future efficacious vaccines and new immune-modulated HIV antivirals. The researchers have now reached one important milestone toward achieving this goal in an international research collaboration with Dr Jenny Ting, University of North Carolina at Chapel Hill, NC, USA, and Dr Sumit Chanda, Sanford-Burnham Prebys Medical Discovery Institute, San Diego, CA. They have identified NLRX1 (nucleotide-binding oligomerization domain, leucine rich repeat containing X1) as an important factor. This protein belongs to the NOD-like receptor family (NOD-like receptors, NLR) of pattern recognition receptors. It acts as a fine-tuning regulator, which can deactivate the early warning system of the immune system. In a high-throughput procedure, NLRX1 had already been identified by König and colleagues as one out of 295 potential proteins important for HIV-1 replication [1]. The fact that it does play a central role, and which mechanisms it uses, has so far been unknown.
König and her international research partners provided evidence that NLRX1 enables HIV-1 infection in immune cells such as macrophages and dendritic cells (cells of the human immune system). The research team was able to show that NLRX1 attenuates the innate immune system by binding STING (stimulator of interferon genes). STING, an important factor in combating viruses, acts as a mediator. After (virus) DNA has been sensed by the receptor cGAS in the cytoplasm of the cells, STING binds to the factor TBK1 (TANK-binding kinase 1) and activates it. Subsequently, type-I interferon and pro-inflammatory cytokines are produced. Moreover, the binding of STING to TBK1 leads to the activation of interferon-stimulating genes (ISG), which prevent the insertion of virus DNA into the nucleus of the cell, thus inhibiting its replication. These defense mechanisms are deactivated when NLRX1 binds to STING. By silencing NLRX, the researchers were able to show that this protein does indeed play a key role, because the cytokine response was considerably increased, thus inhibiting import of the virus DNA into the nucleus.
Therefore, NLRX1 is an attractive target structure for the development of therapies for the treatment of HIV-1. Advanced therapeutics, which block NLRX1 could also be used to act as adjuvants enhancing the efficacy of vaccines. “We believe that our research results can speed up the development of such active substances against HIV-1, which contribute to enhancing the innate immune response to HIV-1”, said König, explaining the significance of these research results.
Original Publication
Guo H, König R, Deng M, Riess M, Mo J, Zhang L, Petrucelli A, Yoh SM, Barefood B, Ventevogel M, Sempowski GD, Zhang A, Colberg-Poley AM, Feng H, Lemon SM, Liu Y, Zhang Y, Wen H, Zhang Z, Damania B, Tsao LC, Wang Q, Su L, Duncan JA, Chanda SK, Ting JPY (2016): NLRX1 Sequesters STING to Negatively Regulate the Interferon Response, Thereby Facilitating the Replication of HIV-1 and DNA Viruses.
Cell Host Microbe. Volume 19, Issue 4, p515–528
DOI: http://dx.doi.org/10.1016/j.chom.2016.03.001
[1] König R, Zhou Y, Elleder D, Diamond TL, Bonamy GMC, Irelan JT, Chiang C, Tu BP, De Jesus PD, Lilley CE, Seidel S, Opaluch AM, Caldwell J, Weitzman MD, Kuhen KL, Bandyopadhyay S, Ideker T, Orth A, Miraglia LJ, Bushman FD, Young JA, Chanda SK (2008): Global analysis of host-pathogen interactions that regulate early-stage HIV-1 replication.
Cell 135: 49-60.
Source: The Paul-Ehrlich-Institut
22.04.2016