Trans-rectal quantitative Shear Wave Elastrography
Prostate cancer is the 2nd leading cause of cancer death in men. It is also the most common diagnosed malignancy in men with near 190.000 new cases in the USA in 2008. Despite the larger use of biological tests (such as prostate specific antigen (PSA)) and imaging modalities (trans-rectal ultrasonography (TRUS) and Magnetic Resonance Imaging (MRI)), there is a slight increase in the annual death rate while the mortality remains stable.
Due to the limited sensibility of TRUS (60 to 70%), a normal examination should not delay the systematic biopsies if digital rectal examination or PSA are abnormal.
The purpose of our study was to evaluate the feasibility of trans-rectal quantitative Shear Wave Elastography (SWE) for prostate cancer evaluation.
Methods and Materials
21 patients presenting with increased PSA values (4-10 ng/mL) were prospectively enrolled after signing an informed consent form. The prostate was studied using transrectal ultrasound (TRUS) with spatial compounded B-mode, colour Doppler US (CDUS) and SWE on the Aixplorer system (Supersonic Imagine, Aix-en-Provence, France; transducer SE12-3). Elasticity measurements and ratios between nodules and adjacent parenchyma were calculated.
Contrast-enhanced US (CEUS) was performed using low MI pulse subtraction after injection of 4.8-9.6 ml of SonoVue® (Bracco, Milan, Italy) using an Aplio XG system (Toshiba MS, Nasu, Japan). Imaging findings were correlated to sextant prostate biopsies (n=12) and targeted biopsies on suspicious areas (n=2-6) detected at SWE and CEUS. MRI with axial and coronal T2w acquisition, axial T1w acquisition, diffusion and dynamic contrast-enhanced MRI (Signa 1.5T and Discovery MR450, GE HealthCare, Milwaukee, WI, USA) was also evaluated. The blinded analysis of the SWE acquisition was performed retrospectively from cineloops and frames. Normal and abnormal patterns were matched with pathology
results.
Results
In 3 patients, it was not possible to localize normal and abnormal patterns using SWE due to a technical problem during the export of the data. The analysis was performed from the remaining 18 patients (mean age 65 ± 6 years, min 54 - max 79). The PSA values were 6.9 ± 2.2 ng/mL.
Eight patients exhibited significant prostate adenocarcinomas. Among the 26 nodules detected either at US or at pathology, 10 were adenocarcinomas with Gleason score above 6 and 16 were adenomatous hyperplasia or focal prostatitis. The correlation between B-mode + pathology and CDUS, CEUS or MRI could not be obtained for 3 / 3 /4 nodules, respectively.
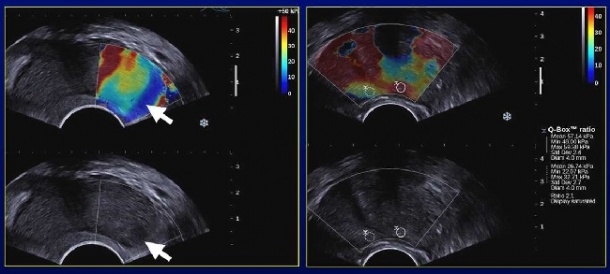
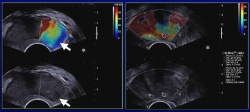
SWE was feasible in all patients. Signals were obtained in both the peripheral and the transition zones with good correlation to anatomical areas. Because of higher attenuation, the deepest transition zone was not correctly assessed over 4-5 cm. Macro-calcifications exhibited very high stiffness values.
Transition zone appeared heterogeneous with stiffness values above 40 kPa. SWE signals were adequate for all peripheral nodules. Limitations resulted from gas interposition and higher pressure of the end-fire transducer on the rectal wall and the
adjacent posterior gland. Prostate cancer nodules exhibited a high stiffness (mean 55 ± 45 kPa, min 23/ max 180 kPa) than the adjacent peripheral gland (mean 18 ± 9 kPa).
Peripheral adenomatous hyperplasia and focal prostatitis exhibited a significantly lower stiffness (mean 19 ± 5 kPa, min 12/ max 28 kPa; p< 0.01). The stiffness ratio between nodule and adjacent parenchyma was significantly higher for cancer (3.0 ± 1.0) compared to benign nodules (1.0 ± 0.20; p< 0.01). The cut-off value of 1.5 allowed the best discrimination of the two populations.
The sensitivity (Se), specificity (Spe), Positive predictive value (PPV) and Negative predictive value (NPV) were calculated for each modality: CDUS, CEUS, SWE and MRI.
CDUS: Se= 50%; Spe= 73%; PPV= 50%; NPV= 73%
CEUS: Se= 67%; Spe= 86%; PPV= 75%; NPV= 80%
SWE: Se= 90%; Spe= 100%; PPV= 100%; NPV= 94%
MRI: Se= 62%; Spe= 78%; PPV= 62%; NPV= 78%
07.03.2011