Tracking the source of heart failure
To treat heart problems, common sense says we should look to the heart. New European research based on an advanced ultrasound system suggests that stiffening of the arteries plays a key role
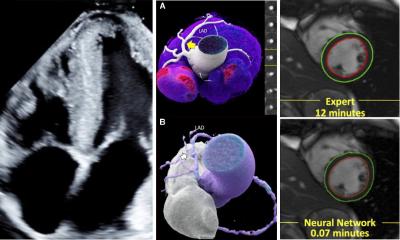
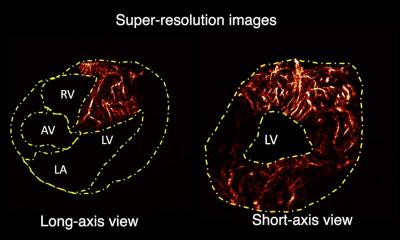
You can die from heart failure even though your heart seems to be pumping normally. You can take pills proven to reduce blood pressure, yet your cardiologist says you are still in danger of a stroke. With these critical conditions reaching epidemic proportions in an aging population, cardiologists are searching everywhere for answers to these mysteries. Shifting their investigation away from the hard-working muscle at the heart of the problem, researchers worldwide in recent years have shown the complex coupling of the heart with stiff large arteries can shape the progress of heart disease. Increased computing power has enhanced the ability of ultrasound systems to measure in a non-invasive way the a relationship between these arteries and a breakdown in the cardio-vascular performance. On the leading edge of this investigation is the new eTRACKING system from Hitachi Aloka that for the first time can measure waves transmitted in the blood moving away from the heart in large arteries, and equally important, the speed and timing of the waves being reflected back by stiff arteries. According to Prof. Alan Fraser of the Cardiff University School of Medicine, who led a collaborative investigation among 14 European centers with eTRACKING, cardiologists can now use these new tools to evaluate and follow the progress of a patient’s condition.
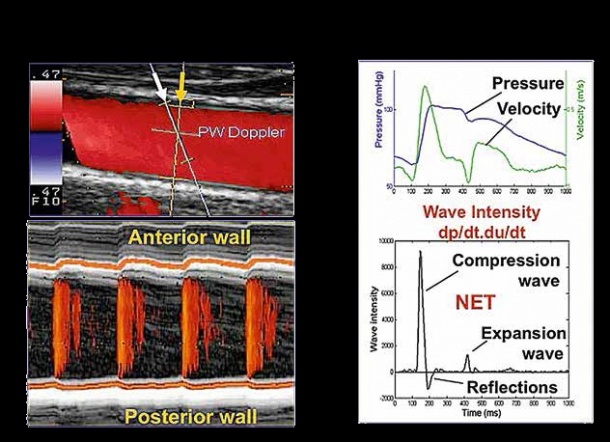
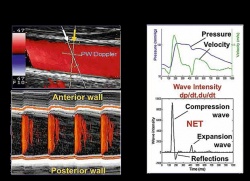
“Expanding what we can measure to assess arterial function, there is a potential to tailor more precisely treatment for individual patients,” he said, adding, “insights from studies using eTRACKING may provide evidence for using one drug over another that we haven’t had previously.” The first applications of eTRACKING will likely be in testing which drugs are most effective in lowering blood pressure and preventing heart failure, while further collaborative studies will help to identify applications for routine clinical practice. What is new with this system, he said, is an ability to look beyond local arterial stiffness to measure waves in the blood circulation being reflected back to the heart. In healthy individuals, as the heart ejects blood, the aorta expands. Thanks to its elastic quality, the vessel recoils which ensures that intermittent emptying from the heart is converted into continuous flow into the peripheral arteries. When the aorta and other main arteries stiffen, the gushing ejection is not absorbed by a stretching action, causing the speed of the waves in the blood to increase. Striking against more hard walls, and moving faster than normal, means that in the periphery there are stronger reflections of waves back to the heart. These backward waves can be two or three times faster than when the vessels were younger and more supple. Pressure in an aging circulatory system increases. With waves coming back before the heart has finished emptying for one pulse, the heart must work harder on the next pulse. The eTRACKING tools enable clinical researchers for the first time using a standard commercially available ultrasound system to measure this complex interplay, using wave intensity. Analysis in large arteries in different regions of the body is possible for artery stiffness, and for the velocity and amplitude of forwards travelling waves and their reflections.
These measurements in the carotid arteries were the focus for the E-tracking International Collaboration (ETIC) study coordinated by Prof. Fraser, which is now awaiting publication. Over 2,000 patients were evaluated, a unusually high population for a study utlizing an new diagnostic imaging system. More surprising is that they were healthy people, individuals without any reported cardiovascular disease, between the ages of three years and 85 years. ETIC is the result of an uncommon partnership between industry and the medical community. The study was funded with an unrestricted grant from Hitachi Aloka with the goal of establishing what is “normal” in the general population before focusing on patients with cardio-vascular disease. The study expands the cardiology community knowledge in what is relatively new territory.
“Many people are working in this field now,” said Prof. Fraser. “Research is always a collaborative effort, so it would not be correct to claim that any one group has done something that no one else has done, or that it is independent of the work of others.” What is unusual is the approach taken by Hitachi Aloka in contributing to the investigation, he said. “The company has been very sensible in asking clinicians, the users of this system, to provide guidance to other clinicians, who will use the instrument, as to what the results mean,” said Prof. Fraser. More often, he said, manufacturers release new technologies and leave users to figure out what it means, whether it has any clinical relevance. “Here physicists and clinicians worked hand-in-hand at a far earlier stage, during the development process, so that the hardware and the software have been developed in a way that we can use it,” he said.
As the chair of last year’s conference on medical devices for the European Society of Cardiology, and the lead author of the society’s position paper on new technologies, Prof. Fraser said the ETIC study provides an example of how industry needs to take up its responsibility in the development of new diagnostic tools.
PROFILE
A Fellow of the Royal College of Physicians as well as the European Society of Cardiology, Alan Fraser is a Professor in Cardiology at the School of Medicine of Cardiff University in Wales. He has earned an international reputation as the head of the Echocardiography Research Group at the Wales Heart Research Institute where he has explored the pathophysiology of heart. He currently is a leading voice in discussions of European regulatory reform as it affects cardiovascular devices, having chaired an ESC Policy Conference and authored the society’s policy statement.
02.11.2012