Telemedicine brings new ways to monitor lung diseases
Aipermon GmbH & Co. KG reports that cooperations with medical device manufacturers Vitalograph and PARI Pharma have opened up new approaches to telemonitor lung patients.
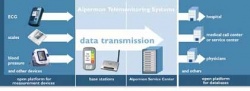
This year, the systems developed by Aipermon were extended to various other medical devices used for diagnosing and treating lung conditions, and are currently being tested in medical studies. ‘With regularly acquired data, collected over a long period of time, medical science hopes to understand better the factors affecting the disease and to find approaches for improving the intervention and medication,’ the firm points out.
To monitor COPD, Aipermon has integrated the Vitalograph asma-1 peak flow meter into the telemetry platform: ‘Data from the peak flow meter can now be transferred fully automatically to a mobile medical assistant (PDA with special software equipment) and from there to a medical care centre. The Pneumology Department at Charité, Berlin uses this technology to prepare a study that enables long-term monitoring of COPD patients in terms of lung function, physical activity (using the 6-minute walk test) and their own self-assessment.’
Correlated with climate data, scientists hope to find clues about the impact on the disease by atmospheric conditions (climate change). Starting in 2011, the study is scheduled for two years and will include approx. 220 patients.
Continuous compliance monitoring
Aipermon technology, combined with eFlow nebulisers by PARI Pharma, enables physicians to check whether the prescribed daily inhalation treatment for cystic fibrosis patients is implemented correctly and regularly. ‘The fully-automatic device signals the date and time, duration and correct completion of each inhalation treatment. With the collection of this data, physicians using such a system, hope for a more successful treatment of cystic fibrosis patients, particularly children, whose parents can receive counselling and support with remote monitoring.
Further telemonitoring system approaches for diseases such as cardiac insufficiency, diabetes, obesity, and high blood pressure will be demonstrated at the Aipermon stand.
Hall 15. Stand C39
18.11.2010