Shocked by the Shock trials?
No need to ask who was really shocked by the Halle Shock trial, followed by the multi-centre Shock II trial.
It was the cardiologists, of course – and heart surgeons – but mainly the manufacturers. Several million Euros worth of lost sales are no small matter. Ah, but, that’s not the end of the story. European Hospital correspondent Holger Zorn observes the emergence from a state of shock
Almost exactly a decade ago, Dr Roland Prondzinsky, then a consultant at the Martin Luther University Hospital in Halle, Germany, prospectively randomised 45 Patients with Acute Myocardial Infarction (AMI) complicated by Cardiogenic Shock (CS), who had to undergo Percutaneous Coronary Intervention (PCI) with or without an additional Intra-aortic Balloon Pump (IABP). He had no idea of the repercussions this would present. In 2010 he published his results: ‘The addition of IABP to standard therapy did not result in a significant improvement in Multi-Organ Dysfunction Syndrome’ and ‘… mechanical support was associated only with modest effects on reduction of APACHE II score as a marker of severity of disease’ [Crit Care Med. 2010; 38:152-60].
Although he observed some haemodynamic improvements, these were not significantly better than those achieved by medication. Conclusion: ‘The use and recommendation for IABP treatment in CS remain unclear.’ [Shock. 2012; 37:378-84].
An unclear recommendation despite the fact, up to that point, that the IABP had a Class I indication in the relevant guidelines for the treatment of cardiogenic shock – a result like this had to be checked. From June 2009 to March 2012, 600 patients with CS-AMI, who were due to undergo PCI, were prospectively, multi-centrically randomised into the IABP group, or the control group, in 37 German hospitals for the Shock II trial.
The study’s endpoint was 30-day survival, and here again: ‘At 30 days, 119 patients in the IABP group (39.7%) and 123 patients in the control group (41.3%) had died’ - no significant difference [NEJM 2012; 367:1287-96]. The one-year results confirmed: ‘Of 595 patients completing 12-month follow-up, 155 (52%) of 299 patients in the IABP group and 152 (51%) of 296 patients in the control group had died. IABP did not reduce 12-month all-cause mortality’ [Lancet 2013; 382:1638-45].
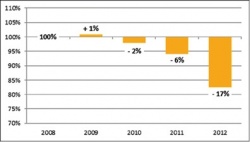
The No Growth disaster
Several well publicised studies, a downgrade in the German-Austrian S3-guidelines on the treatment of CS-AMI to the lower status of a ‘can’ recommendation [Dtsch Arztebl Int 2012; 109:343–51], all this led to a slow and then increasingly faster decline of usage in these two countries (see table).
Not only the authors of the studies quoted above talked about the end of the IABP; European Hospital reported about this extensively (EUR HOSP Vol. 22 issue 4/2012 page 20-21). Still, the world does not seem that impressed. From the month of the Shock II trial results publication to now, another ten studies on the IABP have been published. There are also some new, large studies.
Dr Ludhmila Abrahão Hajjar, Professor for Cardiology at the University of Sao Paulo, will study the effect of counterpulsation on 180 patients due to undergo heart surgery. Dr Nico Pijls, Professor for Cardiology at the Catherina Ziekenhuis Hospital in Eindhoven, the Netherlands, will recruit 100 patients, to find out if a potential persistent ischaemia following PCI can be alleviated with the help of an IABP-implantation. And Dr Philippe Grieshaber, heart surgeon at the University Hospitals in Giessen and Marburg, plans to prophylactically, pre-operatively fit 856 Patients with an IABP for surgical high-risk revascularisation, to see if the clinical outcome, and first and foremost the 30-day-mortality, can be further improved. The state of shock in the community seems to have been overcome for now.
02.09.2014