© MedUni-Wien / LBI Trauma
Article • Implantation of cartilage or stem cells
Remarkable cartilage regeneration
Researchers in Vienna have found a way to regenerate joint cartilage with cartilage matrix from biomaterial.
Report: Michael Krassnitzer
Joint cartilage usually regenerates very poorly. The matrix of cartilage (scaffolding) contains very few cells in deep layers. Moreover, joint cartilage is highly isolated: there are neither regenerative cells in the immediate vicinity that could migrate into the site of the defect and trigger repair, nor vessels that could transport regenerative cells to the cartilage. Unsurprisingly, researchers all over the world are experimenting with different approaches to transporting biomaterials to the joint to make cartilage repair more robust.

Photo © MedUni Wien/feelimage
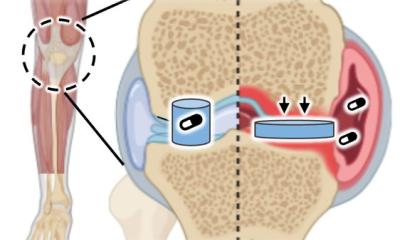
In pre-clinical trials a research team at the Medical University Vienna (Department of Orthopaedics and Trauma Surgery, Division of Trauma Surgery) could achieve remarkably extensive regeneration of joint cartilage from cartilage matrix as biomaterial. The matrix used was harvested from surgery patients who had consented to have their tissue examined for this study. All cartilage cells were removed from the donor matrix leaving native cartilage tissue consisting of collagen type II fibres und glycosaminoglycans. This material, called CartiScaff, is transported to the defective sites in the joint, to create an ideal environment for cartilage cells or stem cells.
The researchers published their findings in the Lancet journal EBioMedicine.
‘The challenge here is to insert native cells into the tissue,’ explains Private Docent Dr Sylvia Nürnberger, the project leader. ‘Cartilage cells are indispensable for cartilage repair,’ she pointed out, because cartilage cells build collagen type II and have to respond to different types of stress. Cartilage matrix without cells would suffer from wear and tear over time. ‘Cartilage cells keep the cartilage intact,’ she explained. ‘They are the maintenance crew of the cartilage.’
Laser opens the door for helper cells
With the biomaterial being so similar to native cartilage, it can stay inside the defect as a part of the regenerated tissues. Alternatively, it can degrade and be replaced with new matrix
Sylvia Nürnberger
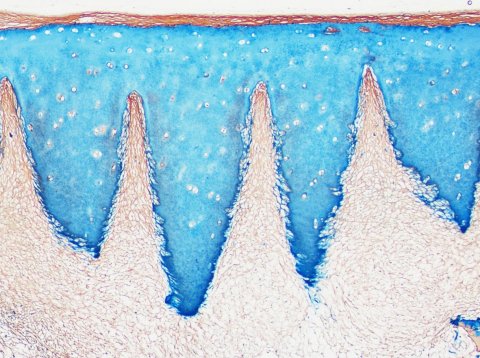
Laser beams have long been a surgical tool, e.g. to correct pathologies in the ear or eye. Nürnberger’s team perforate the cartilage matrix with a laser beam to create an opening for the cells. Tiny grooves were engraved in the matrix, with high precision and all equal in size and distance. These served as access for the regenerative cells, which could thus migrate even in deeper tissue layers. There are three ways to transport the cells to the matrix: either a hole is drilled to the bone marrow and the stem cells can enter the site of the defect, or the matrix and bone marrow harvested from the iliac crest are inserted into the joint in one go. The stem cells then develop into cartilage cells. The third option: the matrix is seeded with stem cells or cartilage cells prior to implantation.
© MedUni-Wien / LBI Trauma
The laser incisions offer another advantage: they provide the cartilage tissue with a high degree of flexibility so it can be more easily fitted onto the rounded surface of the joint bone – comparable to the flexibility of a concertina, Nürnberger observed. ‘The cartilage tissue that grows in the grooves is of rather good quality considering that it’s regenerated tissue.’ Collagen II fibres are orientated perpendicular to the bone and, since the surface is created by the implanted biomaterial, it has the same structure as native cartilage. ‘With the biomaterial being so similar to native cartilage, it can stay inside the defect as a part of the regenerated tissues. Alternatively, it can degrade and be replaced with new matrix,’ Nürnberger explained, adding that follow-up studies will show how the implants develop over time.
Profile:
Dr.rer.nat Sylvia Nürnberger is biologist and head of the working group for Experimental Cartilage Regeneration in the Trauma Surgery Research Laboratory of the Medical University of Vienna and involved in the activities of the Austrian Cluster of Tissue Regeneration. The special focus of her work lies on biomaterials and alternative cell therapies. She developed and patented biomaterials based on natural resources and particularly focuses on translational research to implement fundamental knowledge and developments right down to the clinical area, making them accessible to the medical infrastructure and the patients.
18.02.2021