Image source: Adobe Stock/Africa Studio
News • Cardiology & Surgery
Heart in a box - First beating-heart transplantation
Using an organ from a donor who underwent cardiac death, Stanford Medicine surgeons transplanted a heart while it was beating—the first time such a procedure has been achieved. Initially performed by Joseph Woo, MD, professor and chair of cardiothoracic surgery, and his team in October, the technique has since been used in adult and pediatric patients five more times by surgeons at Stanford Medicine.
Stopping the heart before implantation can damage the cardiac tissue, so keeping it beating during transplantation avoids further injury to the organ.
"This can be revolutionary," said Woo, the senior author on a study describing the procedure that published in the Journal of Thoracic and Cardiovascular Surgery Techniques (JTCVS Techniques) in March. "It's an exciting time for our entire department. This is a big team of very creative individuals that are willing to push the limits of modern technology and health care."
Solving a cardiac conundrum
In a country where 3,500 people currently await a heart transplant, increasing the pool of healthy donated organs is essential.
Brain death donors have long made up the majority of heart transplants, because with those patients, who have been kept on life support before procurement, it's been easier to keep the organ stabilized and assure its health. But with demand outpacing supply, the medical world has been pushed to seek more novel approaches.
Recent technological advances have allowed for more successful heart transplantation from donors who died by what's known as cardiac or circulatory death, in which the heart has already stopped once, either naturally or because they were discontinued from life support. Such procedures increase the number of hearts available for transplant, but outcomes for the recipients are poorer.
These hearts have traditionally been stopped twice—first at death, then immediately before transplantation, after spending time hooked up to a machine that perfuses them with oxygenated blood.
I asked, 'Why can't we sew it in while it is still beating?'
Joseph Woo
"Stopping the heart a second time, just before transplanting, induces more injury," Woo said. "I asked, 'Why can't we sew it in while it is still beating?'"
The Stanford team believed that avoiding a second stoppage of the muscle would improve the quality of the donated heart, but figuring out how presented a challenge.
"It took a little boldness—because it's a challenging, technical thing to do," said Aravind Krishnan, MD, cardiothoracic surgery resident and the lead author on the paper describing the team's findings.
John MacArthur, MD, assistant professor of cardiothoracic surgery, and Brandon Guenthart, MD, clinical assistant professor of cardiothoracic surgery, have also used the technique on adults. Michael Ma, MD, assistant professor of cardiothoracic surgery, recently undertook the first pediatric case.
"By showing that it's translatable it means it can change the entire community," said Woo, the Norman E. Shumway Professor of cardiothoracic surgery.
Added Guenthart, "There's excitement around the clinical aspect of it. We're doing a lot of work in the lab to study it further and take it beyond what we're doing now."
From box to chest
In brain-death donor transplants, the heart is stopped, removed from the deceased donor and transported on ice to the transplantation center. Though it can take a prolonged amount of time to reawaken the heart within the recipient, it has been stopped only once.
Organs recovered from a donor after cardiac death, on the other hand, experience some degree of oxygen deprivation after the heart has stopped beating, before procurement can begin. And studies have shown that a prolonged period without blood circulation in the heart impairs its performance after transplantation and diminishes survival outcomes.
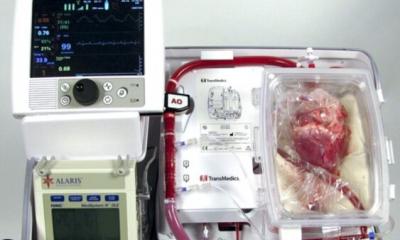
This is why those organs require the aid of a perfusion machine rather than being placed on ice. That organ preservation device—a relatively new invention known as a "heart in a box"—gets it pumping again and perfuses it with warm, oxygenated blood while it's being transferred.
The Stanford patients showed improved outcomes on the implantation end, leaving the hospital sooner than usual because the heart and its new host meshed more quickly.
Keeping the heart beating really seems to make a difference in heart strength with less time spent on the heart-lung machine.
John MacArthur
"We thought that stopping the heart a second time was enough to weaken it," MacArthur said. "Keeping the heart beating really seems to make a difference in heart strength with less time spent on the heart-lung machine."
Ma, who used the procedure on the first pediatric patient earlier this month, said the gains on the front end—continuous function of the organ leading to a shorter time spent in the hospital by the recipient—are immediate. And improvement in long-term outcomes for the organ and its new host are inevitable.
"We won't be able to prove that for a long time, but there are enumerable benefits from this technique," he said. "I was beyond excited to use this procedure pioneered by Joe and his team. It was a really rewarding experience."
Recommended article
News • Donation after cardiac death
'Heart in a box' technology gives hope to transplant patients
A new heart transplant technique, which allows surgeons to transplant donor hearts that have stopped beating after death, is reducing waiting lists for patients in Australia and potentially around the world.
The beat goes on
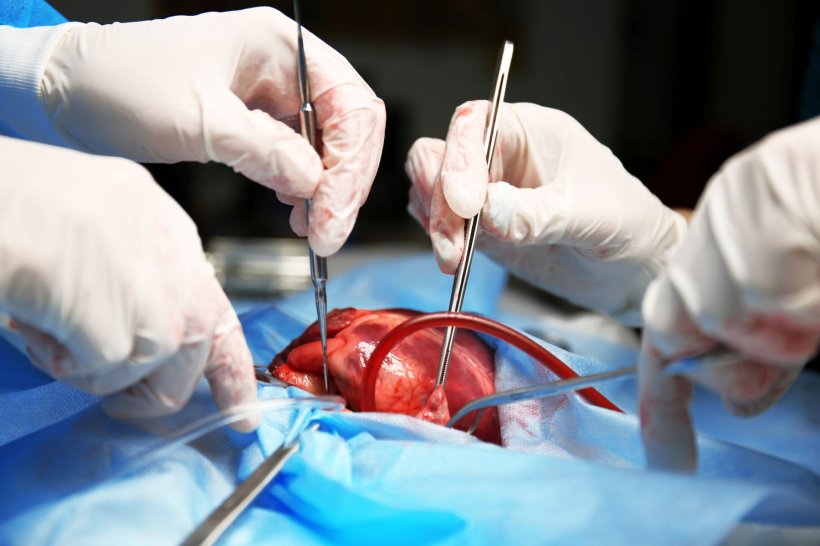
The first beating heart transplant, carried out by Woo and surgical team members Chawannuch Ruaengsri, MD, clinical assistant professor of cardiothoracic surgery, along with Krishnan and Patpilai Kasinpila, MD, resident in cardiothoracic surgery, lasted four hours.
On that October day, the Stanford Health Care team was ready to receive a heart procured from a cardiac-death donor and transported via the heart in a box.
The team quickly transitioned the heart to a cardiopulmonary bypass machine already supporting the patient. The machine, which pumps blood and oxygen throughout the body, ensured the uninterrupted flow of warm blood. Woo then began the challenging process of sewing a beating heart into the recipient.
Krishnan said the team took comfort in knowing the machine was there as a backup if it proved too difficult. But it didn't, and the Stanford Health Care team looked on as the heart kept its rhythm.
"It was beating so well," Ruaengsri said.
All six patients are doing well. And the department is excited about continuing to push the boundaries of what's possible.
"In order to do this, we had to challenge dogma," said Woo, whose predecessor Norman E. Shumway performed the first adult heart transplant in the U.S. in 1968 at Stanford Hospital. "That's how innovations occur."
Added Guenthart, "We're looking into ways of never having to stop the heart—that's the next step."
Source: Stanford University Medical Center
24.04.2023