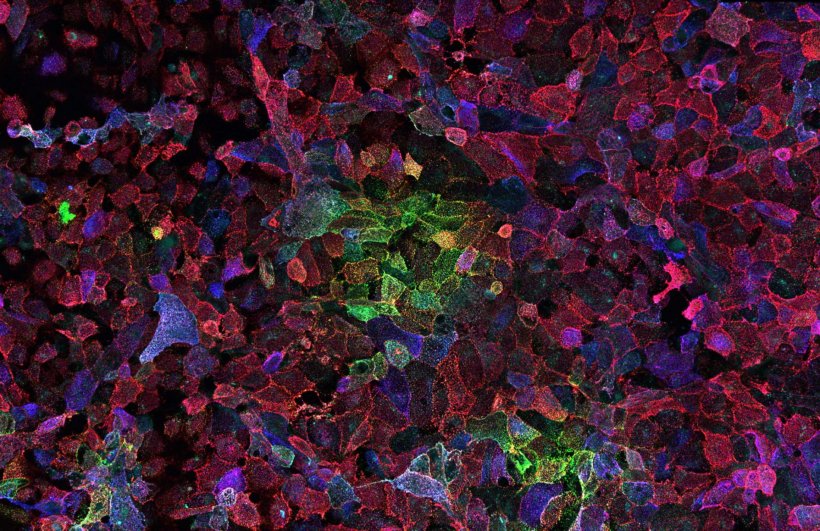
© Alberto Domingo Lopez-Munoz, Laboratory of Viral Diseases, NIAID/NIH
News • Infection in vaccinated patients
How coronavirus variant Omicron BA.1 remodels immune memory
Teams from the internal medicine department of the Henri-Mondor AP-HP hospital, the Institut Necker – Enfants Malades, the Mondor Institute for Biomedical Research, the Institut Pasteur, Inserm, and the Paris-Est Créteil University studied immune memory after infection with the Omicron BA.1 variant in patients vaccinated with three doses of the messenger RNA Covid-19 vaccine.
The results of this study (MEMO-VOC), coordinated by Dr Pascal Chappert and Pr Matthieu Mahévas, in collaboration with Dr Pierre Bruhns and Dr Félix Rey were published in the journal Immunity.
The Spike protein of Sars-CoV-2 Omicron BA.1 carries 32 mutations compared to the ancestral strain (Hu-1) originally identified. These mutations significantly alter neutralizing antibodies induced by natural Sars-CoV-2 infection and/or vaccination with an encoding mRNA vaccine.
Immune memory is a mechanism that protects individuals against reinfection. This defense strategy of the body, which is the basis of the success of vaccines, includes the production of protective antibodies in the blood (detected by serology) as well as the formation of memory cells (memory B lymphocytes*), capable of quickly reactivate into antibody-producing cells upon re-infection. The scientific literature has already shown that the repertoire of memory B cells generated by two or three doses of mRNA vaccines contains neutralizing clones against all variants of Sars-CoV-2 up to Omicron BA.1.
Recommended article
Article • Covid-19
Coronavirus update
Years after the first outbreak and spread of coronavirus Sars-CoV-2, its impact can still be felt in everyday life. Keep up-to-date with the latest research news, political developments, and background information on Covid-19.
The research team studied memory B cells after infection with Sars-CoV-2 Omicron BA.1 in 15 individuals previously vaccinated with three doses of the mRNA Covid-19 vaccine encoding the initial Spike protein of the virus. She followed them up to 6 months after infection with Omicron BA.1 to characterize the response of B lymphocytes, from the early immune reaction to the late onset of long-term memory.
This study reveals that infection with the Omicron BA.1 variant mainly mobilizes memory B cells recognizing common proteins between the initial Spike protein and Omicron BA.1 already present in the repertoire formed after vaccination, but few cells directed against specific BA.1 mutations.
Nevertheless, infection with Omicron BA.1 still induces a reorganization in the memory B cell repertoire without altering its diversity, and an improvement in the overall affinity of the memory B repertoire against the common structures of the Spike encoded in the original vaccine (Spike Hu-1) and that of the Omicron BA.1 variant. This reorganization of the memory repertoire is associated with a significant improvement in the ability to neutralize Omicron BA.1. These results suggest that Omicron BA.1 virus infection in vaccinated patients remodels the memory B cell repertoire and enhances the ability of memory cells to recognize conserved Sars-CoV-2 epitopes and neutralize the virus.
Future vaccine strategies will nevertheless be needed to extend the immune response beyond conserved epitopes to deal with future antigenic variations of Sars-CoV-2.
* Immune cells produced mainly in the lymph nodes and spleen following an infection. They persist for a long time in these regions and retain the memory of the infectious agent. If the body is confronted with them again, these cells are immediately mobilized and quickly reactivate the immune system for effective protection of the individual.
Source: Inserm
17.08.2023





