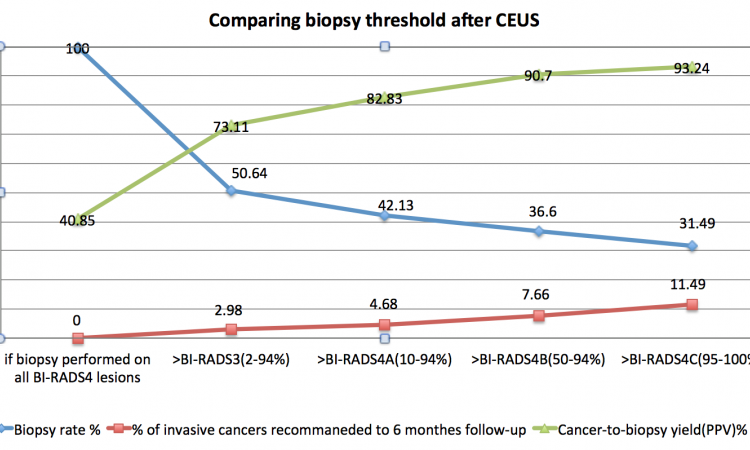
Contrast agents
MultiHance receives approval for use in MRI of the whole body
Bracco Imaging S.p.A., a global leading company in the diagnostic imaging business, today announced the approval of the use of MultiHance® (gadobenate dimeglumine) in Magnetic Resonance Imaging (MRI) of the whole body in adults and paediatric patients (> 2 years). The approval was obtained through a Mutual Recognition Procedure, with the Medicines and Healthcare products Regulatory Agency (MHRA) of the United Kingdom as the Reference Member State, and included Austria, Belgium, Denmark, Finland, France, Germany, Greece, Ireland, Italy, Luxembourg, Netherlands, Norway, Spain and Sweden.
MultiHance®, a paramagnetic contrast agent, was already approved in the European Union for use in MRI of the Central Nervous System (CNS) in adults and paediatric patients older than 2 years of age, and in MRI of the liver, breast and Magnetic Resonance Angiography (MRA) of renal and aorto-ilio-femoral occlusive diseases in adult patients. The new indication wording in the Summary of product characteristics (SmPC) of MultiHance® is now the following:
- MRI of the brain and spine in adults and children above the age of 2 years, where it improves the detection of lesions and provides diagnostic information additional to that obtained with unenhanced MRI;
- MR imaging of the whole body in adults and children (above the age of 2 years) including head and neck region, thoracic space (including the heart and female breast), abdomen (pancreas and liver), abdomen (gastrointestinal tract), retroperitoneal space (kidney, adrenal glands), pelvis (prostate, bladder and uterus) and musculoskeletal system where it facilitates identification of abnormal structures or lesions and helps in differentiating normal from pathological tissues;
- Magnetic Resonance Angiography (MRA) for the assessment of stenoses, occlusions and collaterals in adults and children (above the age of 2 years);
- Specific applications in the heart include measurement of myocardial perfusion under pharmacological stress conditions and viability diagnostics (“delayed enhancement”).
“We are pleased with this approval of a wide range of new and important clinical indications for the highrelaxivityagent MultiHance®, which is based on a thorough assessment of extensive clinical evidence obtained both in adult and paediatric patients over 2 years of age” said Alberto Spinazzi, MD, Senior Vice President, Head of Global Medical and Regulatory Affairs, Bracco Group. “The expanded labeling of MultiHance®, together with its proven contrast-enhancement efficacy and established safety profile, may foster its adoption at hospitals and clinics of the European countries where this approval was obtained” added Fulvio Renoldi Bracco, Head of Global Business Unit Imaging, Bracco Imaging. “The successful outcome of this regulatory procedure further reinforces Bracco’s commitment in providing a comprehensive portfolio of solutions to the healthcare community in the MRI segment.”
Source: Bracco
22.09.2016