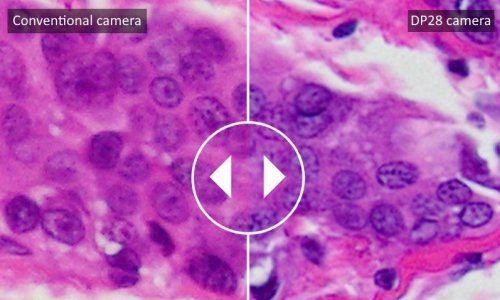
Olympus Analyser most reliable
It runs and runs and runs The Olympus AU640 clinical chemistry analyser is the most reliable analyser considering a period of 12 months. This is the result published in the NHS Purchasing and Supply Agency Analyser Monitoring Programme Annual Report 2006-2007.

The comparitive NHS study assesses the reliability of any mid to high throughput biochemistry and immunoassay analyser, as well as the support provided by the instrument suppliers. The AU640, once again shared top ranking on the survey. The AU640 had a mean monthly downtime of just 6 hours, compared to 25 hours downtime of some analysers registered on the Analyser Monitoring Programme.
As evidenced by the AU640 in the new report, the reliability and quality of support for all Olympus analysers is paramount.
"Once again, we are extremely pleased with the results of this latest report," said Kerri Breasley, Olympus Commercial Marketing Manager, Clinical Chemistry. "The recently launched Olympus AU680 is now set to continue these high standards laid down by the AU640, and to become the latest member of 'The Olympus family heritage group' renowned for reliability and quality."
Source: NHS Purchasing and Supplies Agency (PASA) Centre for Evidence-based Purchasing (CEP) Analyser Monitoring Programme. Annual report 2006-7 (CEP 08001). www.pasa.nhs.uk/cep.
NHS Purchasing and Supplies Agency
NHS PASA CEP evaluates medical technologies and the evidence that supports their use from the perspective of technical, clinical and cost effectiveness to enable healthcare professionals to make sound investment decisions. The full Analyser Monitoring Programme report (CEP 08001) is available at http://www.pasa.nhs.uk/cep.
13.08.2008