Article • Pathology
Fusing microscopy and mass spectrometry
Pathology does not appear to have much in common with satellites, but the concept that satellites combine spatial resolution and image quality will be the future of disease diagnosis, according to researchers.
Report: Lisa Chamoff
Richard Caprioli directs the Mass Spectrometry Research Centre at Vanderbilt University School of Medicine.

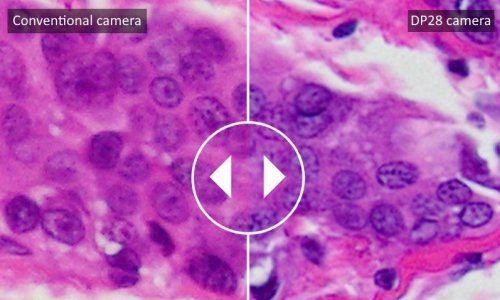
Scientists at Vanderbilt University, Nashville, Tennessee, are combining microscopy and mass spectrometry, using mathematical regression analysis to take advantage of each mobility’s strength. ‘In a satellite imaging process a black and white picture of the ground is taken because black and white cameras have high spatial resolution,’ explains Richard Caprioli, Director of the Mass Spectrometry Research Centre at Vanderbilt University School of Medicine. ‘A colour camera also takes pictures at the same time. The colour camera has millions of colors, but the resolution is lower. A process called image fusion takes the best of both worlds and combines them mathematically to produce an image that has both high resolution and lots of colours.’
Whilst a microscope gets a much higher resolution, it provides no molecular data. Mass spectrometry, on the other hand, is used to measure the molecular characteristics of a disease that are present in the patient’s sample. ‘Instead of just one protein, we are looking at a whole suite of proteins, which we call a disease signature,’ Caprioli explains. ‘Now, when you put together high resolution microscopy and the high molecular dimensionality of mass spectrometry, you produce high-resolution molecular spectroscopy images.’
Caprioli’s team has been studying image fusion to diagnose melanomas, prostate cancer and breast cancer. They are currently in the discovery process, looking at how image fusion differentiates between two stages of a cancer among a couple of hundred patients. The researchers look for a list of proteins that come with the specific disease stage.
Caprioli believes that image fusion for pathology will be used in the next two to three years for the ‘most important cases where the pathologists don’t have the tools for such comprehensive molecular interrogation of tissue.’
Bringing a new technology over the translation bridge is not as easy as many people think.
Prof. Richard Caprioli
In a paper published last year in the journal Nature Methods, Caprioli’s team used image fusion to study protein, peptide, lipid, small metabolite and drug distributions in tissue. ‘Image fusion enables a new multi-modality paradigm for tissue exploration whereby mining relationships between different imaging sensors yields novel imaging modalities that combine and surpass what can be gleaned from the individual technologies alone,’ the authors wrote.
A similar technology has been under development at the Oak Ridge National Laboratory (ORNL) in Oak Ridge, Tennessee, although there it is used for materials characterisation rather than disease progression. At ORNL, the two imaging modalities are being put together mechanically, while researchers at Vanderbilt are using lasers.
The challenge will come in making pathologists aware of the new technology when it will be ready to be put to use and engaging them in discussion. ‘Bringing a new technology over the translation bridge is not as easy as many people think', Caprioli comments.
The College of American Pathologists has identified imaging mass spectrometry as an important technology and has begun its certification process. Additionally, this will eventually need clearance from the USA’s Food and Drug Administration. ‘I believe that, in the coming years, all diagnoses will be done this way, because it makes sense,’ Caprioli confirms. ‘You’re looking at what is wrong with the patient directly and not just picking out a few surrogate markers for the disease. Pathologists have tools that they use today. We are offering a new set of tools that have high molecular dimensionality.’
15.12.2016