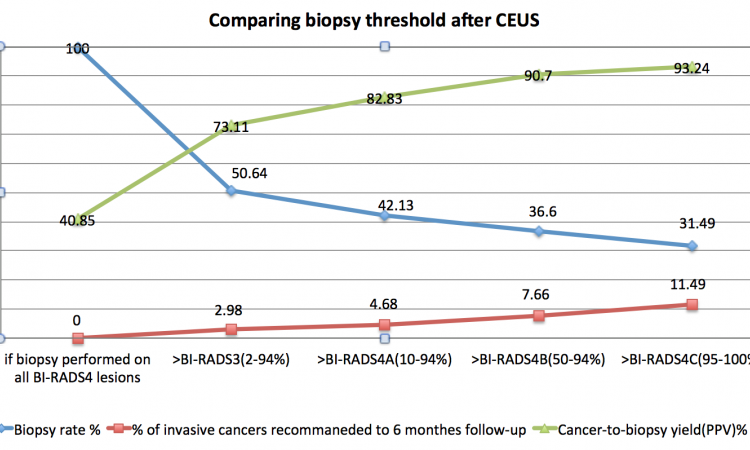
Contraindications removed
Ultrasound Contrast Agent SonoVue usable in case of recent acute coronary syndrome or clinically unstable ischaemic cardiac disease.
Bracco Imaging, a global company in diagnostic imaging, announced important changes in the product information of its ultrasound contrast agent SonoVue in the European Union. Following an extensive evaluation performed by the Committee on Human Medicinal Products (CHMP) of the European Medicines Agency (EMA) of the benefits and risks of SonoVue use in critically ill patients, the decision was made to remove the contraindication for use of the product in patients with recent acute coronary syndrome or clinically unstable ischaemic cardiac disease. Therefore, SonoVue can now be used, even if with extreme caution, in patients with conditions like evolving or ongoing myocardial infarction, typical angina at rest within last 7 days, significant worsening of cardiac symptoms within last 7 days, recent coronary artery intervention or other factors suggesting clinical instability (for example, recent deterioration of ECG, laboratory or clinical findings), acute cardiac failure, Class III/IV cardiac failure, or severe rhythm disorders.
In view of the frequent use of the direct-acting inotropic agent dobutamine for stress echocardiographic procedures, the CHMP also recommended to avoid the use of SonoVue in combination with dobutamine in those patients for which the use of this pharmacological stressor is contraindicated. Therefore, the CHMP decided to add the following contraindication to the Product Information of SonoVue: “Sonovue should not be used in combination with dobutamine in patients with conditions suggesting cardiovascular instability where dobutamine is contraindicated”.
SonoVue is a diagnostic imaging agent which provides critical information for the management of patients with cardiac diseases, including those in the critical care setting. Bracco is pleased that the CHMP has determined that SonoVue® has a favorable risk-benefit profile in patients with recent acute coronary syndrome or clinically unstable ischaemic cardiac conditions, i.e., in the patient population for which the use of SonoVue was previously contraindicated. SonoVue® is marketed in the EU since 2001, has been extensively tested in clinical trials, and is well established in clinical routine use, with an estimated exposure of more than 3 million patients.
11.06.2014