Source: Siemens Healthineers
News • COVID-19
Four commercial immunoassay tests for detection of antibodies
Public Health England, in partnership with the University of Oxford, recently conducted a head-to-head evaluation of four commercial immunoassay tests available in the UK and used for the detection of SARS-CoV-2 antibodies. Siemens Healthineers’ laboratory-based total antibody test1 distinguished itself as the only assay tested found to meet both the sensitivity and specificity requirements set out within the Target Product Profile (TPP) for immunoassays by the UK Medicines and Healthcare products Regulatory Agency (MHRA). The test is broadly available globally.
The evaluation was commissioned by the Department of Health and Social Care (DHSC) and conducted by Public Health England, the clinical research team at the University of Oxford and Oxford University Hospitals NHS Foundation Trust. Several commercial antibody tests were appraised over a three-week period in May-June 2020, with a view to assessing respective performance metrics on precision, using a large, well-characterised sample set. Primary data included evaluation of the assays’ sensitivity and specificity, each of which is key to assessing the overall clinical performance of a test.
High-quality antibody tests will be critical to successfully reopening economies across the globe.
Deepak Nath
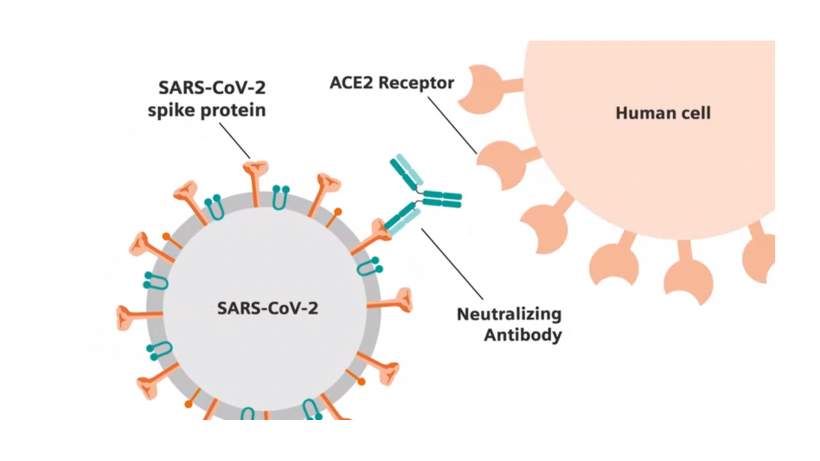
The SARS-CoV-2 total antibody test from Siemens Healthineers is CE marked and has FDA Emergency Use Authorization. The test detects the presence of both IgM and IgG antibodies in blood. This allows for the identification of patients who have developed an adaptive immune response, indicating prior exposure to COVID-19. Critically, the test identifies antibodies to a key protein on the surface of the SARS-CoV-2 virus – a spike protein, which binds the virus to human cells. Multiple potential vaccines in development for SARS-CoV-2 include the spike protein within their focus, raising the possibility that the Siemens Healthineers total antibody test could support the determination of the effectiveness of these vaccines in the future.
“High-quality antibody tests will be critical to successfully reopening economies across the globe. From the start, our mission has been to develop highly accurate tests for SARS-CoV-2 and now we have an external appraisal that confirms our test offers exceptional quality,” said Deepak Nath, PhD, President of Laboratory Diagnostics at Siemens Healthineers. “Laboratories worldwide can be confident that our assay delivers the sensitivity and specificity we promised.”
Source: Siemens Healthineers
Recommended article
News • COVID-19 diagnostics
SARS-CoV-2 IgG antibody test receives FDA Emergency Use Authorization
Beckman Coulter announced that its Access SARS-CoV-2 IgG assay has received Emergency Use Authorization (EUA) from the U.S. Food & Drug Administration (FDA). Beckman Coulter has already shipped tests to more than 400 hospitals, clinics and diagnostics laboratories in the U.S., and has begun distribution of the new antibody test globally to countries that accept the FDA EUA and CE Mark. The…
10.07.2020


