
The Medical Device Regulation (MDR), whose provision on the harmonization of quality standards will become mandatory in 2020, is keeping healthcare professionals in Europe on their toes. At the same time, global consolidation strategies in labs aim to increase effectiveness while reducing service costs. In this issue, we take a closer look at both developments.
Enjoy reading!
Advertisement |
 |
Article • RegulationImplementing MDR is complex and expensive and holds little realityBy 2020 medical devices manufacturers must document the clinical effectiveness of their devices more extensively. The Medical Device Regulation (MDR) presents a fundamental impact on innovation and price calculation for medical devices. Since the ... |
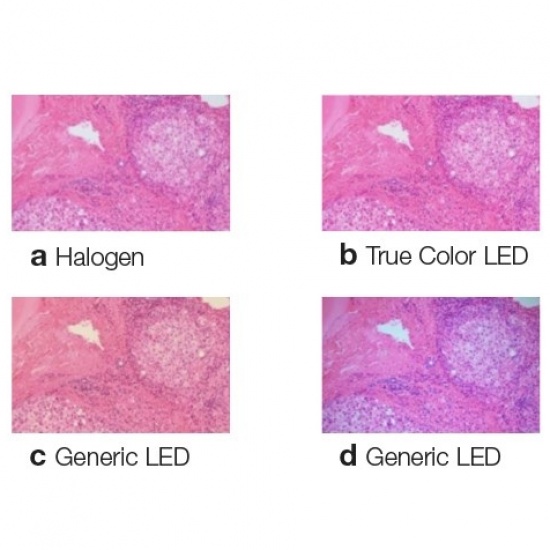 |
Sponsored • IlluminationReliable Diagnoses with Olympus True Color LEDEnsure reliable diagnoses in pathology with Olympus’ market-leading LED technology. True Color LED illumination is a durable, bright light source especially designed to closely match halogen illumination, delivering accurate color reproduction ... |
 |
Article • Economies of scaleLab medicine: Network consolidation continuesLaboratory networks are consolidating across the globe as they seek to deliver a more efficient and cost effective service. The latest developments on several continents were outlined at the FiLM 2018 – Frontiers in Laboratory Medicine congress ... |
 |
Sponsored • Patient safetyXenios hand in hand with hospitalsIn clinical use since 2014, three therapies on one platform, satisfied users and patients. The Xenios console has established itself. All nice and well, one could think and be satisfied with the development and the market-launch. However, those, who ... |
 |
Interview • Clinical practiceLC-MS/MS enters the medical laboratoryOver the last four years, Dr Thomas Stimpfl and his team have integrated mass spectrometry into routine analysis. The analytical technique liquid chromatography–mass spectrometry (LC-MS) combines the physical separation capabilities of liquid ... |
 |
Article • AutomationMass spectrometry is advancing laboratory practiceMass spectrometry is moving laboratory medicine to increasingly automated discrete analysis methods, resulting in ever faster and more reliable results. It is also leveraging economies of scale as an increasingly cost-effective tool, says Craig ... |
 |
Article • MedTech Summit and MT-ConnectMeeting up with Europe’s med-tech and health expertsThe international MedTech Summit and MT-CONNECT, an international medical technology exhibition (Nuremberg, 11-12 April 2018) is a key event in Europe. For many years, developments in digitisation and personalised health have been among core ... |
You are receiving this email because you subscribed to our newsletter on healthcare-in-europe If you don’t want to receive this newsletter anymore, click here to unsubscribe. Keep up-to-date on the latest news from all hospital-related fields! Copyright © 2024 mgo fachverlage GmbH & Co. KG. E.-C.-Baumann-Straße 5, 95326 Kulmbach, Germany |

