News • Life expectancy
Mole-rats might teach us how to become old and healthy
Researchers find novel genes for longevity in mammals
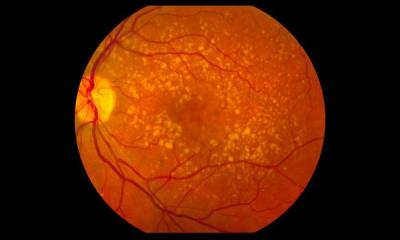
Humans and other mammals have a lot in common. Depending on race, 80 to 98% of mammalian genetic makeup is identical to the human one. Nevertheless, the variety of life expectancy among mammals is huge; and so far, it was unclear, which impact the genetic makeup has on a species’ life span. In rodents, differences in life expectancy and morbidity during aging are particularly high: Despite close relationships with regard to genetic aspects, small rodents like mice or rats live no longer than two to three years, whereas mole-rats or chinchillas have an average life span of 20 to 30 years while staying comparatively healthy. This unique range of life expectancy makes rodents a perfect model system for analyzing the genetic impact of genes on life span and aging in mammals.
Towards this, researchers from the Leibniz Institute on Aging – Fritz Lipmann Institute (FLI) in Jena, Germany – together with colleagues from the Leibniz Institute for Zoo and Wildlife Research (IZW) in Berlin, and the Institute of General Zoology at University Duisburg-Essen – have compared genetic data from 17 rodent species. The team of researchers found 250 genes that were cumulatively under-activated in long-lived rodents, but over-activated in short-lived species. Hence, the researchers concluded that these genes might have been positively selected by evolution for a long and healthy life. Among them, novel genes like RHEBL1, AMHR2, PSMG1, or AGER were identified that have never before been linked to general aging processes and lifespan, but are known to directly contribute to the development of aging-associated diseases like cancer or Alzheimer’s disease in humans. Insofar, the results are also relevant for humans: 85% of the long-lived rodents’ genes are identical to human DNA, but in contrast to humans that are rather prone to illness, long-lived rodents have a very low risk to come down with age-related diseases. The study’s results have now been published in journal PLoS Genetics.
What are the longevity genes responsible for?

For many rodent species, genome and gene expression data are already available. This is why conclusions can be drawn from the recent study, which and how body functions in long-lived rodents differ from the ones in their short-lived relatives. Like this, the researchers from Jena, Berlin and Duisburg-Essen were able to conclude that lifespan extension in rodents can be attributed to changes in their defense against free radicals, iron homeostasis as well as cellular respiration and translation as central parts of the growth program. This confirms aging theories assuming a tradeoff between fast growth and long lifespan: Those genes that make an organism mature fast, contribute to an accelerated aging and a short lifespan later-on, given that they cannot be completely de-activated after maturation and, thus, harm the adult organism. In contrast, in long-lived rodents, these genes are kept under-activated during growth, leading to a slower maturation but also to a longer life.
There are several reasons, why – in the course of billions of years of evolution – a genome might be forced to differentiate and adapt to different environments. Therefore, the identification of longevity genes is only a first step towards the conclusion of hypothetical interrelations, which have to be further analyzed in follow-up studies. However, since the novel genes that were identified, are known to lead to aging-associated diseases in humans, their further analysis seems to be promising for developing new approaches to understand and possibly cure these diseases and to contribute to a long life and healthy aging in humans – in a way, long-lived rodents do.
Source: Leibniz Institute on Aging – Fritz Lipmann Institute (FLI)
04.04.2018