News • Experimental medicine
Loophole in chronic lymphocytic leukemia treatment detected
A team of researchers in Italy and Austria has determined that a drug approved to treat chronic lymphocytic leukemia (CLL) may be less effective in a particular subset of patients.
The study, published in the Journal of Experimental Medicine, reveals that ibrutinib has a diminished capacity to delocalize and kill tumor cells expressing an adhesive protein called CD49d, but combining ibrutinib treatment with drugs that block CD49d activation could prevent the tumor cells from sheltering in lymphoid organs. CLL is the most common leukemia in adults, and it is characterized by the presence of cancerous B cells that accumulate in the lymph nodes, spleen, and liver. Ibrutinib reallocates CLL cells from lymph nodes into the blood by inhibiting Bruton’s tyrosine kinase (BTK), a key enzyme in the B cell receptor (BCR) signaling pathway.
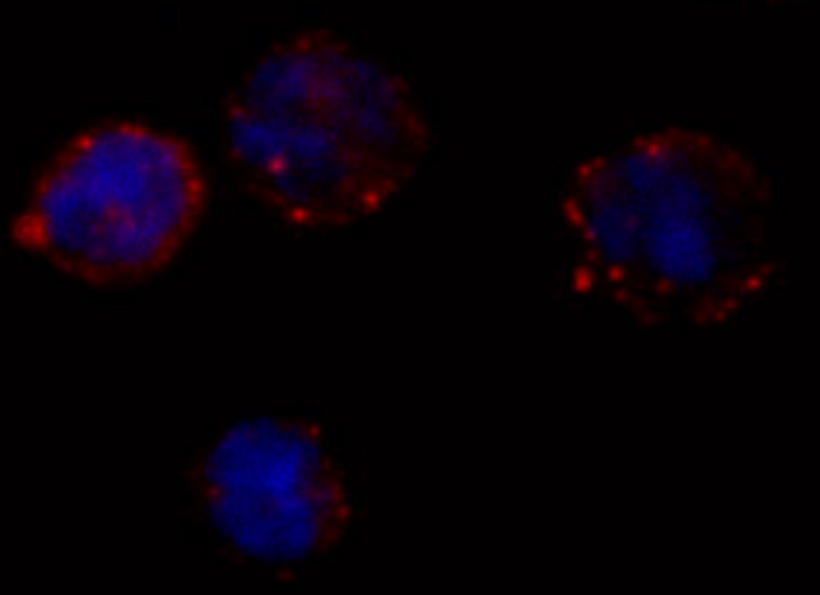
BCR signaling promotes the survival and differentiation of normal, healthy B cells in several ways, including by activating the adhesion receptor VLA-4, which attaches B cells to other, supportive cells within lymph nodes. One of the subunits of VLA-4, CD49d, is highly expressed in about 40% of CLL patients. These patients tend to have poorer outcomes than patients that do not express CD49d, but the role of VLA-4 in CLL is unclear. A team of researchers led by Antonella Zucchetto and Valter Gattei of the CRO Aviano National Cancer Institute in Italy and Tanja Nicole Hartmann of the Paracelsus Medical University in Salzburg, Austria, found that BCR signaling can activate VLA-4 in CD49d-expressing CLL cells, thereby enhancing the cells’ adhesiveness. Even though ibrutinib treatment impaired BCR signaling in these cells, it was unable to fully hinder the pathway from activating VLA-4 and enhancing cell adhesion.
The researchers analyzed three different cohorts of CLL patients from Italy and the United States. In all three groups, patients expressing higher levels of CD49d showed reduced responses to ibrutinib treatment: the drug appeared to be less able to displace tumor cells from lymph nodes into the blood, resulting in decreased lymph node shrinkage and shorter progression-free survival times. “Our results suggest that VLA-4–expressing CLL cells residing in the secondary lymphoid organs can receive BCR-mediated stimuli that can activate VLA-4 even in the presence of ibrutinib,” says Zucchetto. “This activation leads to enhanced retention of VLA-4–positive CLL cells in tissue sites, thereby affecting patient outcome.”
In addition to the ibrutinib target BTK, BCR signaling can proceed through an alternative enzyme called phosphatidylinositide 3-kinase. The researchers found that simultaneously inhibiting both BTK and phosphatidylinositide 3-kinase completely blocked VLA-4 activation in CLL cells. “Our data suggest that evaluation of CD49d expression in patients initiating ibrutinib therapy may identify those cases that would benefit from combination therapy approaches designed to completely block VLA-4 activation and VLA-4–mediated retention of leukemic cells in protective tissue compartments,” says Gattei.
Source: The Rockefeller University Press
05.01.2018